Convert all of the kinematic viscosity data in Table 2.5 for ISO viscosity grades from mm 2
Question:
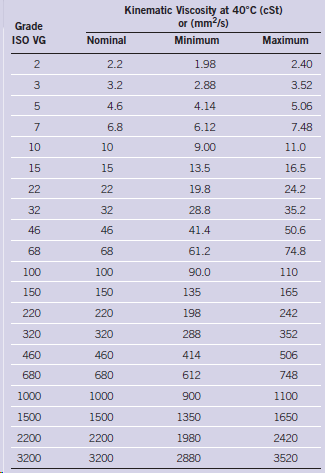
Transcribed Image Text:
Kinematic Viscosity at 40°C (cSt) or (mm2/s) Grade IS VG Nominal Minimum Maximum 2.2 1.98 2.40 3 3.2 2.88 3.52 4.6 4.14 5.06 6.8 6.12 7.48 10 10 9.00 11.0 15 15 13.5 16.5 24.2 22 22 19.8 32 32 28.8 35.2 46 46 41.4 50.6 68 68 61.2 74.8 100 100 90.0 110 150 150 135 165 220 220 198 242 320 320 288 352 460 460 414 506 680 680 612 748 1000 1000 900 1100 1500 1500 1350 1650 2420 2200 2200 1980 3200 2880 3200 3520 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
Convert kinematic viscosity for ISO grades from mm 2 s to SUS First four values ...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At what temperature is the kinematic viscosity of glycerin the same as the kinematic viscosity of helium?
-
State the standard units for kinematic viscosity in the SI system.
-
State the standard units for kinematic viscosity in the U.S. Customary System.
-
Make use of table 1.4 to express the following quantities in BG units: (a) 14.2 km, (b) 8.14 N/m 3 , (c) 1.61 kg/m 3 , (d) 0.0320 N m/s (e) 5.67 mm/hr.
-
Prepare a formal income statement from the partial worksheet for Wright Co. in Figure 12.16. WRIGHT co. PARTIAL WORKSHEET FOR YEAR ENDED DECEMBER 31, 201x Income Statement Account Titles 41000 2 3000...
-
Shelton Engineering completed the following transactions in the month of June. a. Shana Shelton, the owner, invested $105,000 cash, office equipment with a value of $6,000, and $45,000 of drafting...
-
What is a statute of limitations? What is the statute of limitations for negligence actions in California?
-
A municipality expects to use a landfill evenly throughout the 25 years from January 1, 2017, to December 31, 2041. Upon closing the landfill it estimates that it will incur closing costs of...
-
In the xy-plane, what is the y-intercept of the graph of the equation y = (x-2)?
-
Foxy Investigative Services is an investigative services firm that is owned and operated by Shirley Vickers. On November 30, 2018, the end of the fiscal year, the accountant for Foxy Investigative...
-
An oil is tested using a Saybolt viscometer and its viscosity is 526 SUS at 40C. Determine the kinematic viscosity of the oil in mm 2 /s at that temperature.
-
Convert 1600 square millimeters to square meters.
-
(a) Describe the molecule xenon trioxide, XeO3, using four possible Lewis structures, one each with zero, one, two, or three Xe-O double bonds. (b) Do any of these resonance structures satisfy the...
-
Should U.S. national forests become privatized (sold to private owners)? Why or why not?
-
In the United States, a principal responsibility for preserving endangered species (e.g., a pair of endangered birds that chooses to nest on private land) and the costs of exercising that...
-
By applying modern technology to agriculture, the United States has become the most productive food-producing nation in the world. The secret to solving the world food security problem lies in...
-
What are the income-distribution consequences of fashion? Can the need to be seen driving a new car by the rich be a boon to those with lower incomes who will ultimately purchase a better,...
-
In your hometown what system is used to price the publicly supplied water? Why was that pricing system chosen? Would you recommend an alternative?
-
What are the advantages and the disadvantages of purchasing an outlet from a small franchisor?
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Two of the compounds from Problem 13.14 can be prepared from the reaction between a Grignard reagent and an ester. Identify those two compounds, and explain why the other four compounds cannot be...
-
Predict the major organic product for each of the following reactions. a. b. c. d. e. f. ? PCC C-Clz - - NazCrz07 H2SO4. H20
-
Propose a synthesis for each of the following transformations. a. b. c. d. Br
-
At January 1, 2024, M. E. Gainor Corporation had outstanding the following securities: 600 million common shares 20 million 6% cumulative preferred shares, $50 par 6.4% convertible bonds, $2,000...
-
market rate of interest. Interest is payable annually on December 31, Carr Corporation issued $45,000 of 5 percent, 9-year bonds on January 1, Year 1, for a price that reflected a 6 percent Note: Use...
-
Italian Stallion has the following transactions during the year related to stockholders' equity. February 1 Issues 5,400 shares of no-par common stock for $16 per share. May 15 Issues 600 shares of...

Study smarter with the SolutionInn App


