Determine the pressure at the bottom of the tank in Fig. 3.25. 1.2 m --- --- --
Question:
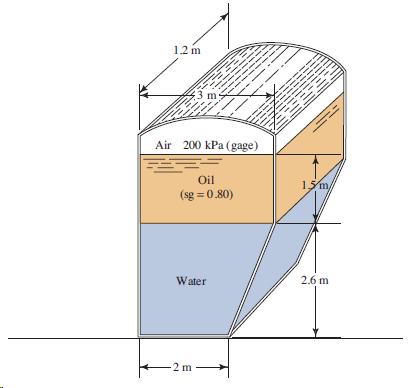
1.2 m --- --- -- -- Air 200 kPa (gage) Oil 15m/ (sg = 0.80) 2.6 m Water 2 m
Step by Step Answer:
p tot p air o h o w ...View the full answer



Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Sciences questions
-
A closed tank contains a liquid (sp. gr. = 0. 80) under pressure. The pressure gauge depicted in Figure P2.2.7 registers a pressure of 4.50 Ã 104 N/m2 (pascals). Determine the pressure at the...
-
The liquid in the open-tube manometer in Fig. 14.9a is mercury, y, = 3.00 cm, and y, = 7.00 cm. Atmospheric pressure is 980 millibars. (a) What is the absolute pressure at the bottom of the V-shaped...
-
A closed rigid tank contains carbon-dioxide at 10 MPa and 100oC. It is cooled until its temperature reaches 0oC. Determine the pressure at the final state (p2). Use (a) The RG model with the...
-
As a security architect for a medium-sized firm, briefly describe how would you approach the following case problems: 1. Sending encrypted email to third parties. 2. Encrypting sensitive data inside...
-
Raymond Company's trial balance at December 31, 2017, is presented below. All 2017 transactions have been recorded except for the items described below and on page 473. Unrecorded transactions: 1. On...
-
The graph below shows the change in pressure as the temperature increases for a 1-mol sample of a gas confined to a 1-L container. The four plots correspond to an ideal gas and three real gases: CO 2...
-
Gruner Company accumulates the following data concerning a proposed capital investment: cash cost \($280,000\), annual cash inflow \($50,000\), present value factor of cash inflows for 10 years 5.65...
-
Given the estimated demand function Equation 2.2 for avocados, Q = 104 40p + 20pt + 0.01Y, use algebra (or calculus) to show how the demand curve shifts as per capita income, Y, increases by $ 1,000...
-
Exhibit 14-8 Plazzi, Inc. sold $400,000 of its 9%, five-year bonds dated January 1, 2013, on May 1, 2013, for $393,000 plus accrued interest. Interest is paid on January 1 and July 1 and...
-
In your opinion, could U.S. complaints about China's policies toward its auto exports actually harm U.S. producers more than they help? Should the U.S. dismiss its complaint with the WTO?
-
Figure 3.24 shows a closed container holding water and oil. Air at 34 kPa below atmospheric pressure is above the oil. Calculate the pressure at the bottom of the container in kPa(gage). 0,25 m Air...
-
Water is in the pipe shown in Fig. 3.26. Calculate the pressure at point A in kPa(gage). Pipe 100 mm - Water 75 mm Mercury (sg = 13.54)
-
Will an increase in the demand for a monopolists product always result in a higher price? Explain. Will an increase in the supply facing a monopsonist buyer always result in a lower price? Explain.
-
For this messaging strategy practice exercise, you'll assume the role of a marketing consultant hired by the Centers for Disease Control and Prevention (CDC). Your job is to design a public health...
-
Poor Mr.Wang He has been struggling with his injuries. The skin on his hands degenerated and required skin grafts. The skin graft on his dominant hand got a staph infection. After 8 months of...
-
Determining Friction from the Stopping Distance 1. The simulation The code below shows what happens to a system that has an initial velocity, and then slides to a stop due to friction. 1. Try...
-
Can you determine whether an investment is warranted based solely upon the NPV? If three mutually exclusive investments all show about the same NPV, how might you determine which to pursue?
-
e. Identify the highest favorable variance and highest unfavorable variance from the six listed in requirement d, and provide one possible cause of each variance.
-
Use a calculator to evaluate the expression. Round your answer to three decimal places. (-8.11) -4
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
Draw a bond-line structure for each of the following compounds: (a) 2-Heptyne (b) 2,2-Dimethyl-4-octyne (c) 3,3-Diethylcyclodecyne
-
Predict the products for each of the following reactions: (a) (b) H2 Lindlar's catalyst :? H2 Pt -? Na NH, (1) ? Ni,B Pd Na NH3 (/)
-
Predict the products for each of the following reactions: H2 Lindlar's catalyst 2 Pt
-
HW 1 Wishbone Multimedia had the following note payable transactions for 2017 and 2018. June 1, 2017 purchased equipment from Texco costing $18,000 by issuing a one-year, 8% note payable. General...
-
Kingbird Corporation reported net income of $253,200 in 2020 and had 197,000 shares of common stock outstanding throughout the year. Also outstanding all year were 42,000 options to purchase common...
-
Fraser Corporation declares a cash dividend to common shareholders. How does this transaction impact the accounting equation: Assets [Select] Liabilities [Select] and, Stockholders' Equity [Select]

Study smarter with the SolutionInn App


