Figure 9.27 shows a duct in which methyl alcohol at 25°C flows at the rate of 3000
Question:
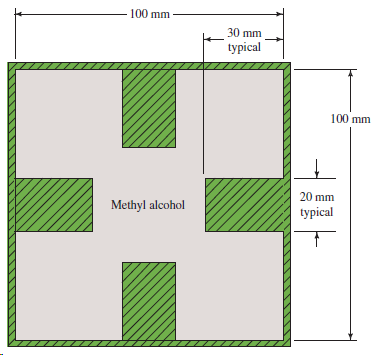
Transcribed Image Text:
100 mm 30 mm typical 100 mm 20 mm Methyl alcohol typical
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
A0100 m 4002003m WP40108003064 m Q3000 L...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A hair dryer is basically a duct in which a few layers of electric resistors are placed. A small fan pulls the air in and forces it through the resistors where it is heated. Air enters a 1200-W hair...
-
Consider conditions for which a mercury-in-glass thermometer of 4-mm diameter is inserted to a length L through the wall of a duct in which air at 77C is flowing. If the stem of the thermometer at...
-
Figure 9.29 shows a system in which methyl alcohol at 77°F flows outside the three tubes while ethyl alcohol at 0°F flows inside the tubes. Compute the volume flow rate of each fluid required...
-
The table shows equivalent temperatures in degrees Celsius and degrees Fahrenheit. (a) Plot the data with Fahrenheit temperature on the x-axis and Celsius temperature on the y-axis. What type of...
-
On June 1, 2014, Novack Company purchases equipment on account from Moleski Manufacturers for $50,000. Novack is unable to pay its account on July 1, 2014, so Moleski agrees to accept a three-month,...
-
Use Table to determine the percent of data specified. Less than z = -0.45 Table of Areas to the Left of z When z Is Negative .00 .01 .02 .03 .04 .05 .06 .07 .08 .09 -3.4 0003 .0003 .0003 .0003 .0003...
-
Assume that 1 year ago, the spot rate of the British pound was $1.70. One year ago, the 1-year futures contract of the British pound exhibited a discount of 6 percent. At that time, you sold futures...
-
Clarkson Computer Company distributes a specialized wrist support that sells for $30. The company's variable costs are $12 per unit; fixed costs total $360,000 each year. Required a. If sales...
-
Entries for Investment in Bonds, Interest, and Sale of Bonds Bocelli Co. purchased $84,000 of 5%, 10-year Sanz County bonds on May 11, Year 1, directly from the county, at their face amount plus...
-
1. What are the advantages and disadvantages of Rovios current business model? 2. Do you agree with the company chairman Kaj Hed when he says he is satisfied with Rovios current situation? Rovio...
-
A furnace heat exchanger has a cross section like that shown in Fig. 9.28. The air flows around the three thin passages in which hot gases flow. The air is at 140°F and has a density of 2.06...
-
In Fig. 9.26, ethylene glycol (sg = 1.10) at 77°F flows around the tubes and inside the rectangular passage. Calculate the volume flow rate of ethylene glycol in gal/min required for the flow to...
-
Determine the bare module, total module, and grassroots cost of the following: Styrene plant described in Appendix B, Project B.3 Figure B.3.1 B.3 STYRENE PRODUCTION, UNIT 400 Styrene is the monomer...
-
The corporate Treasurer for a global technology company is comparing the returns offered by U.S. Treasury bills with the returns for risk-free securities issued by the government of Romania. Both...
-
23 months ago you borrowed $2000.The loan has 24 monthly payments and an interest rate of 0.7% per month.How much interest will you pay in the last month?
-
Explain the meaning of Cost of Quality and the classification of cost of quality?
-
If markets are inefficient, then why have traders not been able to exploit the wealth of arbitrage opportunities that should be out there?If markets are efficient, then what reason would traders have...
-
A particle moves along a straight line with velocity given by the function v(t) = 4t6t + 2t + 5, where t is measured in seconds. Find the total distance traveled by - the particle from t 0 tot 3...
-
Determine the appropriate rotation formulas to use so that the new equation contains no xy-term. 25x 2 36xy + 40y 2 1213x - 813y = 0
-
What is a make-or-buy decision?
-
A sample containing 42.1 g of Ar is enclosed in a container of volume 0.0885 L at 375 K. Calculate P using the ideal gas, van der Waals, and RedlichKwong equations of state. Based on your results,...
-
The experimental critical constants of CH 4 are found in Table 7.2. Use the values of Pc and T c to calculate V c . Assume that CH 4 behaves as a. An ideal gas b. A van der Waals gas c. A...
-
The observed Boyle temperatures of H 2 , N 2 , and CH4 are 110, 327, and 510. K, respectively. Compare these values with those calculated for a van der Waals gas with the appropriate parameters.
-
Assume that Jenny's Beauty Salon, Inc. is organized as an S corporation, and began operation on September 20, 2023. The corporation's required year is the calendar year, but it is qualified to make a...
-
The Balance Sheet equation implies which of the following?Group of answer choicesAssets + Liabilities = Owners EquityAssets + Owners Equity = LiabilitiesAssets = Liabilities + Owners EquityAssets =...
-
Mountainside Marble & Tile has declared a 15% stock dividend. The journal entry on the date of declaration will debit a. Stock Dividends for the market value of the shares to be distributed. b. Stock...

Study smarter with the SolutionInn App


