To what height will the jet of water rise for the conditions shown in Fig. 6.40? p=
Question:
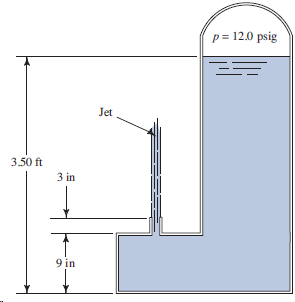
Transcribed Image Text:
p= 12.0 psig Jet 3.50 ft 3 in 9 in
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
h 350 ft 10 ft 250 ft D...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
To what height will the jet of fluid rise for the conditions shown in Fig. 6.39? Jet 2.60 m 75 mm 0.85 m
-
A glass rod of diameter d1 = 1.5 mm is inserted sym- metrically into a glass capillary with inside diameter d2 = 2.0 mm. Then the whole arrangement is vertically oriented and brought in contact with...
-
Water of mass m = 20 g is enclosed in a thermally insulated cylinder at the temperature of 0 C under a weightless piston whose area is S = 410 cm2 . The outside pressure is equal to standard...
-
Select only 2. Two questions which can be used to address ethical issues impacting organizations and Human Resource Management include: What would my Supervisor say? Does this behavior or result meet...
-
Journalize, without explanations, the following transactions of Digbey Auto Parts, a distributor that uses the periodic inventory system, during the month of June 2017: Jun. 3 Purchased $16,800 of...
-
In Section 7.2.2 we introduced the notion of a universal reference type (void * in C) that refers to an object of unknown type. Using such references, implement a poor mans generic queue in C, as...
-
On January 1, 20XA, Rain Oil Corporation bought a developed lease for $300,000. During 20XA, Rain Oil Corporation incurred $600,000 of IDC on a successful well. Reserves of 400,000 barrels were...
-
Read the following letter and help Shady Slim with his tax situation. Assume that his gross income is $172,900 (which consists only of salary) for purposes of this problem. December 31, 2018 To the...
-
The average amount of money spent for lunch per person in the college cafeteria is $7.06 and the standard deviation is $2.49. Suppose that 9 randomly selected lunch patrons are observed. Assume the...
-
Sally Saia operates Double S Riding Academy, Inc. The academys primary sources of revenue are riding fees and lesson fees, which are provided on a cash basis. Sally also boards horses for owners, who...
-
What pressure is required above the water in Fig. 6.12 to cause the jet to rise to 28.0 ft? The water depth is 4.50 ft. h
-
Solve Problem 6.88 using the direct application of torricelli's theorem. What depth of fluid above the outlet nozzle is required to deliver 200 gal/min of water from the tank shown in Fig. 6.37? The...
-
Population Growth. The population P, in thousands, of a senior community is given by Where t is the time, in months. a) Graph the function on the interval [0, (] . b) Find the population at t = 0, 1,...
-
What are the molecular mechanisms of intracellular transport and vesicular trafficking, including the roles of motor proteins, cytoskeletal filaments, and membrane fusion complexes in mediating the...
-
Question 1 (2 points): Suppose Scarlett purchased a 3-year bond with the face value of 20,000 in the primary market. The current risk-free interest rate was 0.5%, and a risk premium on that bond is...
-
The total manufacturing cost for Bolan Hosier House is 302,000 dollars, with the sales 523,610 dollars, while the work in process at the start of the month is amounted 35,600 dollars with the ending...
-
Using the Bohr model, determine the energy, in electron volts, of the photon produced when an electron in a hydrogen atom moves from the orbit with n = 6 to the orbit with n = 1 . ( Assume that the...
-
The GAPS most recents earning per share were $1.75. Analysts forecast next years earnings per share will be $1.82. The appropriate P/E ratio is 17. What is the reasonable price for a share of GAP...
-
A factory produces gasoline engines and diesel engines. Each week the factory is obligated to deliver at least 20 gasoline engines and at least 15 diesel engines. Due to physical limitations,...
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
When used as a solvent, will acetone favor an S N 2 or an S N 1 mechanism? Explain. Acetone
-
Determine whether each of the following reactions proceeds via an S N 1 or S N 2 mechanism and then draw the product(s) of the reaction: (a) (b) (c) (d) (e) (f) .? , Br cP HMPA
-
In Chapter 23, we will learn several methods for making primary amines (RNH 2 ). Each of these methods utilizes a different approach for forming the CN bond. One of these methods, called the Gabriel...
-
Presented below is selected financial information for two divisions of Crane Brewing. Supply the missing information for the lettered items. Contribution margin Controllable margin Average operating...
-
On April 6, 2022, Ben purchased a house to use as residential rental property. He made extensive repairs to the house and had it ready for rent on July 5, 2022. He began to advertise the house for...
-
Due to the sluggish economy, the Wheels Company has experienced some difficulty in selling its bicycles. The following data relate to the current year: Sales 9,000 units x $100@ $900,000 less:...

Study smarter with the SolutionInn App


