In Exercises 1 through 20, find the indicated indefinite integral. 5xe-x2 dx
Question:
In Exercises 1 through 20, find the indicated indefinite integral.
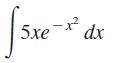
Transcribed Image Text:
5xe-x²2 dx
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
5xex dx 1 Let ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Calculus For Business, Economics And The Social And Life Sciences
ISBN: 9780073532387
11th Brief Edition
Authors: Laurence Hoffmann, Gerald Bradley, David Sobecki, Michael Price
Question Posted:
Students also viewed these Mathematics questions
-
Gofar Travel Vehicles sells new and used recreational vehicles. When new vehicles arrive at Gofar Travel Vehicles from the manufacturer, a new vehicle record is created. Included in the new vehicle...
-
In Exercises 1 through 20, find the indicated indefinite integral. S 3x + 1 dx
-
In Exercises 1 through 20, find the indicated indefinite integral. fax + (x + 1)(2x + x) dx
-
A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major...
-
The July 1, 20X5, trial balance for the Bond Redemption and Interest Debt Service Fund of the County of Hawaii, Hawaii, is presented here. The resources of the fund are committed to debt service. The...
-
Prove that A n is simple for n 5, following the steps given. a. Show A n contains every 3-cycle if n 3. b. Show An is generated by the 3-cycles for n 3. c. Let r and s be fixed elements of {l, 2, ...
-
Which type of bond is unsecured? a. Common bond b. Mortgage bond c. Serial bond d. Debenture bond
-
What activities in managing the pay system are likely candidates to be outsourced? Why?
-
Northam Packaging Company produced 2,06,000 tons of coated paper board in 1989. This paper board was sold to consumer product firms ( processors) who formed the paper board into cartons, then filled...
-
In Exercises 15 through 19, the demand and supply functions, D(q) and S(q), for a particular commodity are given. Specifically, q units of the commodity will be demanded (sold) at a price of p = D(q)...
-
In Exercises 3 through 36, find the indicated integral and check your answer by differentiation. fox. (x + 1)(x + 2x + 5) dx
-
1. Corporate income taxes are payable on profit. How would you describe "profits" that result from the Google "Double-Irish-Dutch" tax strategy? 2. What are some possible changes to the tax code that...
-
Posters.com is a small Internet retailer of high-quality posters. The company has $710,000 in operating assets and fixed expenses of $161,000 per year. With this level of operating assets and fixed...
-
What advanced persuasion techniques can be used in business writing to influence decisions or actions? How do you apply these techniques while maintaining ethical standards ?
-
How do you conduct a comprehensive audience analysis to ensure your business communication resonates with a diverse set of stakeholders? What factors do you consider in tailoring your message to...
-
Silver nitrate has a molar mass of 169.87 g/mol. To make 250 mLof 1% weight/volume AgNO3 in ethanol, what mass of AgNO3 isrequired?
-
The substitution reaction of molybdenum hexacarbonyl, Mo(CO)6 ,with a variety of other molecules L are believed to occur by thefollowing mechananism. Mo(CO)6----> Mo(CO)5+CO Mo(CO)5-----> Mo(CO)5 L...
-
How are CSAs different than FSAs?
-
5. Convert the following ERD to a relational model. SEATING RTABLE Seating ID Nbr of Guests Start TimeDate End TimeDate RTable Nbr RTable Nbr of Seats RTable Rating Uses EMPLOYEE Employee ID Emp...
-
An excess of lymphocytes is consistent with various forms of viral infection, such as hepatitis. What is the probability that a normal adult will have 40 or more lymphocytes? Infectious Disease The...
-
What is the probability a normal adult will have 50 or more lymphocytes? Infectious Disease The differential is a standard measurement made during a blood test. It consists of classifying white blood...
-
How many lymphocytes would have to appear in the differential before you would feel the normal pattern was violated? Infectious Disease The differential is a standard measurement made during a blood...
-
Seneca Inc. is a Canada-based exporting firm that expects to receive payments denominated in both euros and British Pounds (GBP) in one month. Based on today's spot rates, the Canadian dollar value...
-
Explain how volume is used to make buy and sell decisions in Technical Analysis?
-
Assume that Xs Corporation is considering the establishment of a subsidiary in Norway. The initial investment required by the parent is $5,200,000. If the project is undertaken, Xs would terminate...

Study smarter with the SolutionInn App


