a. Name and give the formulae of substances A to E. b. Write balanced chemical equations for
Question:
a. Name and give the formulae of substances A to E.
b. Write balanced chemical equations for the reactions in which compounds B, C and E were formed.
c. Where would you expect to find acid A?
d. Universal indicator solution was added to solution C. What colour did it go?
e. Upon addition of dilute hydrochloric acid to solution C, a neutralisation reaction took place.
(i) Write a balanced chemical equation for the reaction taking place.
(ii) Name the salt produced in this reaction.
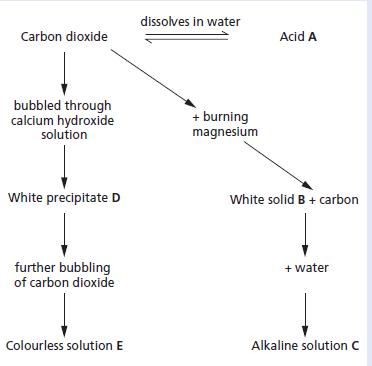
dissolves in water Carbon dioxide Acid A bubbled through calcium hydroxide solution + burning magnesium White precipitate D White solid B + carbon further bubbling of carbon dioxide + water Colourless solution E Alkaline solution C
Step by Step Answer:
a b A HCO3 C MgOH2 E CaHCO32 for B CO g Mg s for C ...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
In what chemical shift ranges would you expect to find the proton NMR signals of ethyl acetate (CH3CO2CH2CH3)?
-
What kind of organizational structure would you expect to find in (a) A fast-food restaurant (b) A company like GE or GM (c) A biotechnology company?
-
Would you expect to find adenineguanine or cytosinethymine base pairs in DNA? Why?
-
Factor each polynomial. 64y 9 + z 6
-
A solution was prepared by dissolving 0.834 g of sulfur, S8, in 100.0 g of acetic acid, HC2H3O2. Calculate the freezing point and boiling point of the solution.
-
The temperature varies linearly from sea level to approximately \(11 \mathrm{~km}\) altitude in the standard atmosphere. Evaluate the lapse rate- the rate of decrease of temperature with altitude-in...
-
Presented below are selected financial data for two competitors. Discuss why the return on assets increased for Company A but decreased for Company B. Company A. Company B Return on Assets Return on...
-
Gerald/Brooke, Ltd. manufactures shirts, which it sells to customers for embroidering with various slogans and emblems. The standard cost card for the shirts is as follows. Bobby Brickley, operations...
-
What is the difference between refundable and non-refundable credits? How do credits affect self-employment taxes, net investment income taxes and alternative minimum taxes
-
(a) Show that , in S1 units for B c - Because B c decreases with increasing temperature, the right side is negative. The superconducting phase has the lower entropy: it is the more ordered phase. As ...
-
Limestone is an important raw material used in many different industries. a. One of the properties of limestone is that it reacts with acids. (i) Why do farmers spread powdered limestone on their...
-
The following question is about carbon dioxide. a. Name and give the formula of each of the substances A, B and C. b. Identify by name the different pieces of apparatus D, E, F and G. c. Draw and...
-
The Cutting Department of Sonoras Textiles had the following information about production and costs for the month of July: Beginning work in process, 9,200 units that are 100 percent complete as to...
-
The longest shot on a golf tournament was made by Mike Austin in 1974. The ball went a distance of 471m. Suppose the ball was shot horizontally off a cliff at 80.0 m/s. Calculate the height of the...
-
Below are some common delegation issues with examples. Give your own examples of over-delegation, under-delegation, and refusal to accept a legitimate delegation, and explain what you would do in...
-
Solve 5 x 10 (6 x 4-10) + 20+2 -
-
Jimba Jomba Jumba Juice (JJJJ) is currently selling for $56 per share. The dividends for the next three years are expected to be $1.80 for 2022, $2.00 for 2023, and $2.25 for 2024. We forecast the...
-
what is delegation? please explain the principle of delegation?
-
1. What three reasons did the defendant assert to support a request for a new trial? 2. What standard applies to an appellate court's consideration of a contention that a trial court's evidentiary...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
A photon-powered spacecraft of mass 10.0 kg emits radiation of wavelength 225 nm with a power of 1.50 kW entirely in the backward direction. To what speed will it have accelerated after 10.0 y if...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
The work function for metallic cesium is 2.14 eV. Calculate the kinetic energy and the speed of the electrons ejected by light of wavelength (a) 700 nm, (b) 300 nm.
-
a. For this base-case scenario, what is the NPV of the plant to manufacture lightweight trucks? The NPV of the plant to manufacture lightweight trucks, based on the estimated free cash flow is $...
-
Suppose you observe the following situation: Security Pete Corporation Repete Company Beta 1.85 1.54 Expected Return .195 .168 a. Assume these securities are correctly priced. Based on the CAPM, what...
-
Consider two local banks. Bank A has 81 loans outstanding, each for $1.0 million, that it expects will be repaid today. Each loan has a 3% probability of default, in which case the bank is not repaid...

Study smarter with the SolutionInn App


