The apparatus shown below was set up. Give explanations for the following observations. a. The formation of
Question:
The apparatus shown below was set up.
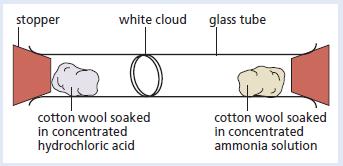
Give explanations for the following observations.
a. The formation of a white cloud.
b. It took a few minutes before the white cloud formed.
c. The white cloud formed further from the cotton wool soaked in ammonia.
d. Cooling the concentrated ammonia and hydrochloric acid before carrying out the experiment increased the time taken for the white cloud to form.
Transcribed Image Text:
stopper white cloud glass tube cotton wool soaked in concentrated hydrochloric acid cotton wool soaked in concentrated ammonia solution
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a the Vapours from the two gases diffuse towards each other and where they meet the...View the full answer

Answered By

Amruta Hajare
I have bachelors degree in general science commonly known as BSC. After teaching vocational colleges for some years I realized my calling to teach was for young minds, giving them strong foundation for future careers and then went back to University and did post graduate diploma in education (PGDE). I am therefore experienced tutor tackling various topics in Biology, chemistry and mathematics under IGCSE curriculum. I give services to my clients at a fair price and relate very well with both parents and students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Concentrated hydrochloric acid contains 1.00 mol HCl dissolved in 3.31 mol H2O. What is the mole fraction of HCl in concentrated hydrochloric acid? What is the molal concentration of HCl?
-
A barium mineral was dissolved in hydrochloric acid to give a solution of barium ion. An excess of potassium sulfate was added to 50.0 mL of the solution, and 1.128 g of barium sulfate precipitate...
-
Concentrated hydrochloric acid is usually available at a concentration of 37.7 percent by mass. What is its molar concentration? (The density of the solution is 1.19 g/mL.)
-
In Exercises find the derivative of the function. y = x(x + 1)
-
A weak acid, HA, is dissolved in water. Which one of the following beakers represents the resulting solution? (Water molecules have been omitted for clarity.)
-
Amber Corporation acquired 60 percent ownership of Sparta Company on January 1, 20X8, at underlying book value. At that date, the fair value of the noncontrolling interest was equal to 40 percent of...
-
In recording credit card sales, when do you debit Accounts Receivable and when do you debit Cash?
-
Coldplay Corporation incurred the following costs while manufacturing its product. Instructions(a) Identify each of the costs as direct materials, direct labor, manufacturing overhead, or period...
-
Points 3 Triangle ABC is congruent to triangle DEF. Find the missing angles and side BC in the congruent triangles. B 65 55 C E 22 cm F OLE=60, A = 58, F = 2C = 60, BC = 21 units OLE = 65, A = 58, F...
-
Dom's Pizza Company uses taste testing and statistical analysis of the data prior to marketing any new product. Consider a study involving three types of crusts (thin, thin with garlic and oregano,...
-
Define the following terms using specific examples to help with your explanation: a. Element b. Metal c. Non-metal d. Compound e. Molecule f. Mixture g. Flocculation h. Gel i. Foam j. Emulsion k. Sol.
-
Use the kinetic theory to explain the following: a. When you take a block of butter out of the fridge, it is quite hard. However, after 15 minutes it is soft enough to spread. b. When you come home...
-
Imagine that an auditor finds an error in the clients financial statements, and that correcting the error in the clients financial records would result in EPS declining by one penny. Why might the...
-
Cowan, an engineer, is the president of Arco Engineering. At a meeting of the board of directors, Cowan was asked to explain why audits of the company are made by (1) internal auditors, (2)...
-
The public accounting profession has achieved remarkable growth and stature in the United States since the beginning of this century, to the point that by the 1960s, some claimed the U.S. was the...
-
Review the discussion of ethical issues and the need theories of motivation. Discuss those issues with the goal of taking a strong position on them.
-
The accounting profession's commitment to achieving high quality in rendering professional services is demonstrated by the breadth and effectiveness of its multilevel regulatory framework. Required...
-
The criticisms raised about behavior modification. Discuss those criticisms. Do those criticisms limit the use of behavior modification in organizations?
-
Research the impact and scope of financial statement fraud on businesses and consumers in the United States in the 21st century. Assess the importance of fraud deterrence and prevention on...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Prove that the perfect gas temperature scale and the thermodynamic temperature scale based on the Second Law of thermodynamics differ from each other by at most a constant numerical factor.
-
Evaluate (ClS/ClV)]' for (a) A van der Waals gas, (b) A Dieterici gas (Table 1.7). For an isothermal expansion, for which kind of gas (and a perfect gas) will /).5be greatest? Explain your conclusion.
-
Two of the four Maxwell relations were derived in the text, but two were not. Complete their derivation by showing that (S/V)T = (p/T)V (T/P)s = (V/S)p
-
Profitability ratios Blank______. Multiple choice question. analyze and compare the earnings and dividends for different sized companies in different industries are used to analyze a company's...
-
To successfully complete this assignment, please submit a word document answering each of the questions below by using both the AICPA Code and your state code. Will your independence be threatened if...
-
- Aa A A S Paragraph Styles Editing Dictate Sensitivity Editor Reuse Files Styles Voice Sensitivity Editor Reuse Files C. Hujusting Accounts Question: Find out the amounts indicated by blanks in...

Study smarter with the SolutionInn App


