The table below shows the melting points, boiling points and densities of substances A to D. a.
Question:
The table below shows the melting points, boiling points and densities of substances A to D.
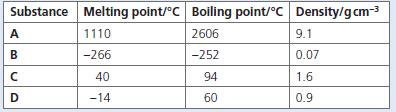
a. Which substance is a gas at room temperature?
b. Which substance is a liquid at room temperature?
c. Which substances are solids at room temperature?
d. Which substance is most likely to be a metal?
e. Which substance will be a liquid at −260 °C?
f. What is the melting point of the least dense nonmetal?
g. Which substances are gases at 72 °C?
Transcribed Image Text:
Substance Melting point/°C Boiling point/°C Density/gcm-3 A 1110 2606 9.1 -266 -252 0.07 40 94 1.6 D -14 60 0.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
Answers According to The International Union of Pure and Applied Chemistry IUPAC the standard room ...View the full answer

Answered By

G Sampath
A Global Online Tutor having 9+ years of experience with a Master’s Degree in Organic Chemistry. Have been tutoring chemistry to the students of different country curriculum's according to their subject needs. Have made many students fall in love with chemistry and delivered high-quality, result-oriented, personalized, one-on-one live video/audio/chat enabled sessions. I have strong theoretical and practical knowledge in all areas of chemistry and the ability to teach the students in an easy and understandable manner.I assure you of providing the highest quality work on any assigned task before the mentioned time.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The table below shows the stock price, earnings per share, and dividends per share for three companies as of October 2007: (a) Determine the price-earnings ratio and dividend yield for the three...
-
The table below shows the stock price, earnings per share, and dividends per share for three companies as of October 2007: a. Determine the price-earnings ratio and dividend yield for the three...
-
The table below shows the closing monthly stock prices for Amazon.com and Google during 2007. Calculate the simple three-month moving average for each month for bothcompanies. 56810 41397 57 19817...
-
Hanley asks his assistant to collect details on those costs included in the $21,000 indirect-cost pool that can be traced to each individual job. After analysis, Wigan is able to reclassify $14,000...
-
Give the conjugate base to each of the following species regarded as acids. a. HSeO4 b. PH4+ c. HS d. HOCl
-
On January 2, 20X8, Total Corporation acquired 75 percent of Ticken Tie Companys outstanding common stock. In exchange for Ticken Ties stock, Total issued bonds payable with a par value of $500,000...
-
Record entries for these transactions assuming the allowance method is used: Jan. 10 The $300 account of customer Cool Jam is determined uncollectible. April 12 Cool Jam unexpectedly pays in full the...
-
A machine part consists of a thin 40.0-cm-Iong bar with small l.15-kg masses fastened by screws to its ends. The screws can support a maximum force of 75.0 N without pulling out. This bar rotates...
-
**#1.) In the diagram below a cylinder with a height of 8 yds and a radius of 13 yds. Determine the surface area a) In terms of the nearest tenth 13 yd 8 yd
-
In Fig P2.11, sensor A reads 1.5 kPa (gage). All fluids are at 20°C. Determine the elevations Z in meters of the liquid levels in the open piezometer tubes B and C. ABC 2 m Air 1.5 m Gasoline 1m...
-
a. How many atoms of the different elements are there in the formulae of the compounds given below? (i) Nitric acid, HNO 3 (ii) Methane, CH 4 (iii) Copper nitrate, Cu(NO 3 ) 2 (iv) Ethanoic acid, CH...
-
Name the method which is most suitable for separating the following: a. Oxygen from liquid air b. Red blood cells from plasma c. Petrol and kerosene from crude oil d. Coffee grains from coffee...
-
The price of gold is currently $1,200 per ounce. The forward price for delivery in one year is $1,300 per ounce. An arbitrageur can borrow money at 3% per annum. What should the arbitrageur do?...
-
What is the difference between a foreseen party relationship and a foreseeable party relationship?
-
a. Who owns and maintains custody of the working papers? b. Is it ever appropriate for the auditor to disclose the contents of working papers to anyone other than the client?
-
How did the Barchris case change the auditors working climate?
-
a. Describe the types of calculations and comparisons commonly used in analytical procedures. b. What premise underlies the use of analytical procedures in auditing? c. Identify five sources of...
-
For each of the following procedures, indicate whether sampling would or would not be used. Give reasons for your answers. a. Examinations of marketable securities. b. Confirmation letters sent to...
-
Novo is a Danish multinational firm that produces industrial enzymes and pharmaceuticals (mostly insulin). In 1977, Novo's management decided to "internationalize" its capital structure and sources...
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
Express the van der Waals equation of state as a virial expansion in powers of 1/Vm and obtain expressions for Band C in terms of the parameters a and b. The expansion you will need is (1- xtI = 1 +...
-
The second virial coefficient B' can be obtained from measurements of the density p of a gas at a series of pressures. Show that the graph of p/ p against p should be a straight line with slope...
-
The following equations of state are occasionally used for approximate calculations on gases: (gas A) p Vm = RT(1 + b/V m)' (gas B) p(V m - b) = RT. Assuming that there were gases that actually...
-
Consider how Star Valley, a popular ski resort, could use capital budgeting to decide whether the $8 million Blizzard Park Lodge expansion would be a good investment. Assur conce assur Click the icon...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Debit Credit Balance 1/1 28,000 Credits Debits 146,000 Balance 12/31 38,000 Manufacturing Overhead...
-
Your company has total receivables outstanding at 31 December 2020 of $20,000. You think that about 2% of these balances will not be collected and wish to make an appropriate allowance. You have not...

Study smarter with the SolutionInn App


