In aerobic respiration, the Krebs cycle is regarded as a series of small steps. One of these
Question:
In aerobic respiration, the Krebs cycle is regarded as a series of small steps. One of these steps is the conversion of succinate to fumarate by an enzyme, succinate dehydrogenase.
a. State the role played by dehydrogenase enzymes in the Krebs cycle and explain briefly the importance of this role in the production of ATP.
b. An investigation was carried out into the effect of different concentrations of aluminium ions on the activity of succinate dehydrogenase. The enzyme concentration and all other conditions were kept constant. The graph below shows the results of this investigation.
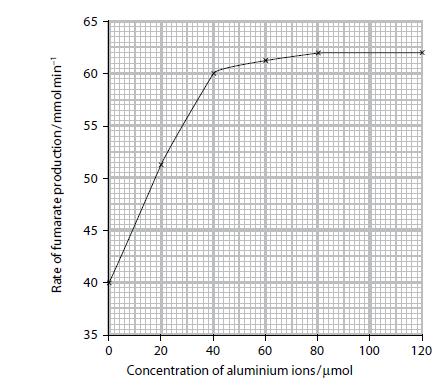
With reference to the graph:
i. Describe the effect of the concentration of aluminium ions on the rate of production of fumarate
ii. Suggest an explanation for this effect.
65 60 55 50 - 45 40 35 20 40 60 80 100 120 Concentration of aluminium ions/umol Rate of fumarate production/mmol min
Step by Step Answer:
A The succinate dehydrogenase enzyme catalyzes the oxidation of succinate into fumarate in the Krebs ...View the full answer



Cambridge International AS And A Level Biology
ISBN: 9781107636828
4th Edition
Authors: Mary Jones, Richard Fosbery, Jennifer Gregory, Dennis Taylor
Students also viewed these Sciences questions
-
A series of small packages being moved by a thin conveyor belt that passes over a 300-mm-radius idler pulley. The belt starts from rest at time t = 0 and its speed increases at a constant rate of 150...
-
A series of small machine components being moved by a conveyor belt passes over a 120-mm-radius idler pulley. At the instant shown, the velocity of point A is 300 mm/s to the left and its...
-
A series of small machine components being moved by a conveyor belt passes over a 120-mm-radius idler pulley. At the instant shown, the angular velocity of the idler pulley is 4 rad/s clockwise....
-
Chambers Corporation purchased a piece of equipment for $36,000. It estimated a 6-year life and $6,000 salvage value. Thus, straight-line depreciation was $5,000 per year [($36,000 $6,000) 6]. At...
-
The Group IIA carbonates decompose when heated. For example, MgCO3(s) MgO(s) + CO2(g) Use enthalpies of formation (see Appendix C) and calculate the heat required to decompose 10.0 g of magnesium...
-
The admission data for freshmen at Tech during the past 10 years are as follows: Techs admission objective is a class of 5,000 entering freshmen, and Tech wants to forecast the percentage of offers...
-
If betas truly are mean-reverting, we would expect them to move back toward: a. -1 b. 0 c. 1 d. +00
-
EKC Company uses the retail inventory method. The following information for 2016 is available: Required: Compute the cost of the ending inventors under each of the following cost flow assumptions...
-
Find the range of possible values for the following measurements, which were rounded to the nearest mm, tenth of m, and hundredth of kg respectively: a 24 mm b 3.2 m c 1.75 kg
-
What are the triads using roman numerals and figured bass numbers for this excerpt? 30-3. Use Roman numerals and figured bass numbers to identify triads in these works. a. Schubert, "Death and the...
-
Copy and complete the following passage describing the adaptations of rice for growing with its roots submerged in water. The stems and leaves of rice plants have very large ............. in tissue...
-
What are the products of the light dependent reactions of photosynthesis? A. ATP, RuBP and reduced NAD B. ATP, oxygen and reduced NADP C. GP, oxygen and reduced NAD D. GP, reduced NADP and RuBP
-
Evaluate each integral using any algebraic method or trigonometric identity you think is appropriate, and then use a substitution to reduce it to a standard form. J-1 V1 + x sin x dx
-
Which of the following is true for Dell, the computer company? a. Most of Dell's costs are for direct materials and direct labor. Indirect costs are a small proportion of total costs. b. Dell uses...
-
The White Flour Company mills wheat into flour. The equivalent units are measured in terms of tons of flour produced. At the beginning of the year, the mill contained 20 tons of flour that was 30...
-
Compare the full absorption and variable incomes when finished goods inventory increases and when it decreases.
-
Dana Helton is considering expanding her business. She plans to hire a salesperson to cover trade show's. Because of compensation, travel expenses, and booth rental, fixed costs for a trade show are...
-
Martha Trent is trying to prepare a personal budget and has identified the following list of monthly costs. Identify each cost as fixed, variable, or mixed. Indicate a possible cost driver for any...
-
Stock is often considered the riskiest type of investment vehicle. The reasoning behind the risky label is the fact that company stock prices can move up or down rapidly, and many internal and...
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
When 1-methylcyclohexene is treated with HCl, a Markovnikov addition is observed. How would you use 1 H NMR spectroscopy to determine that the major product is indeed the Markovnikov product?
-
How many signals will be expected in the 1 H NMR spectrum of each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) `NO2
-
Compare the structures of ethylene, acetylene, and benzene. Each of these compounds produces only one signal in its 1 H NMR spectrum. Arrange these signals in order of increasing chemical shift. -...
-
Futura Company purchases the 79,000 starters that it installs in its standard line of farm tractors from a supplier for the price of $10.00 per unit. Due to a reduction in output, the company now has...
-
Posting a Revenue Journal The revenue journal for Sapling Consulting Inc. follows. The accounts receivable controlling account has a July 1, 20Y2, balance of $880 consisting of an amount due from...
-
Assume a company is preparing a budget for its first two months of operations. During the first and second months it expects credit sales of $40,000 and $73,000, respectively. The company expects to...

Study smarter with the SolutionInn App


