a. What type of polymerisation reaction formed the polymer shown below? b. Draw the displayed formula of
Question:
a. What type of polymerisation reaction formed the polymer shown below?
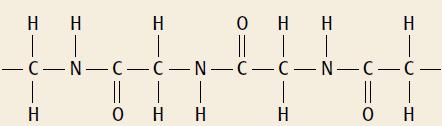
b. Draw the displayed formula of the single monomer used to make the polymer shown in part a.
Transcribed Image Text:
H H H O H H H | -C-N-C-C-N-C-C-N-C-C- H O H H H он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a The polymer shown in above figcan be synthesized by condensatio...View the full answer

Answered By

Sunil Singh
Education:-
1.B.Sc ( chemistry, mathematics, industrial chemistry) , 2017-2020
2.M.Sc ( chemistry), 2020 - continuing.
3.Course on computer course certificate.
4.NPTEL certificate awarded for passing the course industrial inorganic chemistry with 73?.
5.Certificate of predominance:-
The certificate is awarded for passing the online quiz contest Organized by department of chemistry , Arasu college of science and art for women karur-06 with passing score 84?.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
The strongly hydrophilic polymer shown below is used in soft contact lenses. (a) Suggest how you might synthesize this polymer from methacrylic acid and any other reagents you need. (b) What is it...
-
Draw the structure of the monomer or monomers used to synthesize the following polymers: a. b. c. d. e. f. g. h. For each polymer, indicate whether it is a chain-growth polymer or a step-growth...
-
Draw a short segment of the polymer formed from cationic polymerization of 3,3- dimethyloxacyclobutane. CHj CH3 3,3-dimethyloxacyclobutane
-
There are major responsibilities of system administrator as listed below: o Start-up and shut down the system o Performance tuning o Managing user accounts o System security o Backup and recovery o...
-
A business has two investment choices. Alternative 1 requires an immediate outlay of $2000 and offers a return of $7000 after seven years. Alternative 2 requires an immediate outlay of $1800 in...
-
Resistance to change is more likely to succeed if the forces resisting the change are stronger than the forces driving the need for change. Describe some of the specific tactics that resisters would...
-
Disclosure of the fair values of financial instruments (as required by FRS 13 in the UK) is better than measuring them at fair values in the financial statements (as is often required by IAS 39)...
-
Highline Hospital provides a wide range of health services in its community. Highlines board of directors has authorized the following capital expenditures: Intra-aortic balloon pump .. $1,400,000...
-
Amanda and Steve are preparing for a party. Amanda buys flowers from a florist, and Steve buys groceries from a supermarket. Are the two events dependent or independent? Justify the answer. A. B. O...
-
Registration at Southern University has always been a time of emotion, commotion, and lines. Students must move among four stations to complete the trying semiannual process. Last semesters...
-
a. Identify the fragments that would cause peaks in the mass spectrum of HOCH 2 COCH 3 with the following m/e values: i. m/e = 15 ii. m/e = 17 iii. m/e = 31 iv. m/e = 43 v. m/e = 57 vi. m/e = 59 b....
-
a. How does a non-solvent-based adhesive, such as an SMP, set to become a solid? b. What type of monomers react together to form an epoxy resin? c. i. Draw the displayed formula of the monomer used...
-
In Problems 19 26, factor the difference of two squares. 25x 2 4
-
A 75-kg student is on a 15-kg bike. A second student pulls the bike with a force of 38 N. What is the acceleration of the bike and student? If the second student maintains his pull for 8.0 seconds,...
-
Anderson Enterprises manufactures tires for the Formula I motor racing circuit. For August, it budgeted to manufacture and sell 3,900 tires at a variable cost of $76 per tire and total fixed costs of...
-
3. A 3.15-kg steel ball traveling at 25.0/335 meters per second collides with and embeds itself in a 10.5-kg block of clay traveling at 4.20/80 meters per second. Find the velocity of the combined...
-
Imagine we have two boxes with n and n moles of ideal gas each; the boxes are in thermal contact with each other and the combined system is isolated from the world and has a total energy E. Using the...
-
You can run 9.2m/, 20% faster than your brother. a) How much head start should you give him in order to have a tie race over 90m?
-
Why is the earnings yield not usually an adequate measure of the investor's required return on equity?
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
For each pair of compounds below, predict which compound will have the higher boiling point and explain your choice: a) CH 3 CH 2 CH 2 OCH 3 or CH 3 CH 2 CH 2 CH 2 OH b) CH 3 CH 2 CH 2 CH 3 or CH 3...
-
Derive the following expression for calculating the isothermal change in the constant volume heat capacity: (CV/V)T = T (2P/T2)V.
-
For each of the following compounds, identify all groups that would be considered substituents, and then indicate the systematic name as well as the common name for each substituent: (a) (b) (c) (d)...
-
Radko Inc. had the following transactions related to its common and preferred shares: Transactions January 15 Issued 230,000 no par common shares for $33 per share. Issued 1,700 $20 stated value...
-
Pronghorn Ltd. and Culver Ltd. incurred the following merchandise transactions in June. June 10 11 12 19 Pronghorn sold $5,400 of merchandise to Culver, terms n/30, FOB shipping point. The...
-
Hudson Partners provides management consulting services to government and corporate clients. Hudson has two support departments administrative services (AS) and information systems (IS)and two...

Study smarter with the SolutionInn App


