Look at Figure 29.27. a. What is the retention time of the compound shown? b. What is
Question:
Look at Figure 29.27.
a. What is the retention time of the compound shown?
b. What is the approximate relative molecular mass of the compound shown?
c. How would the compound be identified?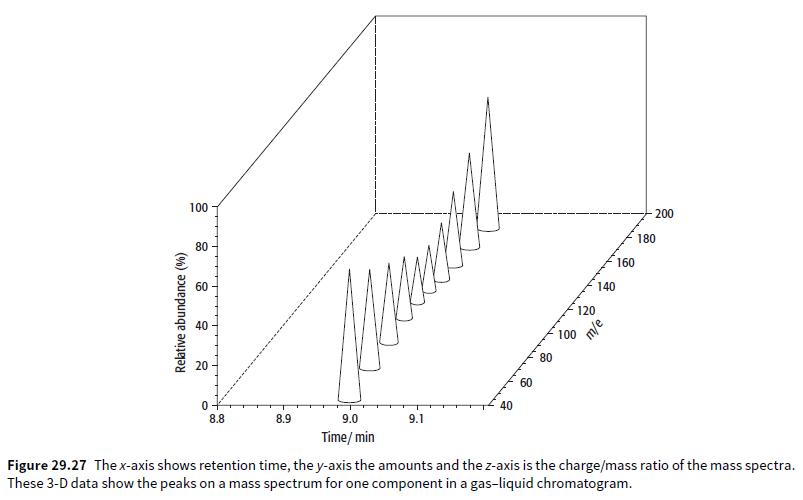
Transcribed Image Text:
100 200 80 180 160 60 140 120 40 100 20 80 60 40 8.8 8.9 9.0 9.1 Time/ min Figure 29.27 Thex-axis shows retention time, the y-axis the amounts and the z-axis is the charge/mass ratio of the mass spectra. These 3-D data show the peaks on a mass spectrum for one component in a gas-liquid chromatogram. Relative abundance (%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
a We have to answer what is the retention time of the compound shown The retention time ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Use this information to compute the following: FunTime Cruiseline offers nightly dinner cruises departing from several cities on the eastern coast of the United States including Charleston,...
-
A large distributor of oil-well drilling equipment operated over the past two years with EOQ policies based on an annual holding cost rate of 28%. Under the EOQ policy, a particular product has been...
-
(a) Make a graph showing the retention time of each peak in Figure 24-25 in chromatograms B, F, and C as a function of position along the line BC. Predict the retention times for solvent compositions...
-
Swish Designs specialises in designing commercial office space in Auckland. The CEO, Ralph Polo has reviewed the financial results and has noticed that operating profits were below budget. He also...
-
The owner of a sporting goods store is considering remodeling the store to carry a larger inventory. The cost of remodeling and additional inventory is $60 000. The expected increase in net profit is...
-
What do buyers look for when they assess the past performance of a business?
-
What are the components of a complete set of LAS 1-compliant financial statements?
-
Strudler Real Estate, Inc., a construction firm financed by both debt and equity, is undertaking a new project. If the project is successful, the value of the firm in one year will be $280 million,...
-
Find an equation of the line L. The equation is Type an equation. Simplify your answer.) 48
-
Edmund and Lydia Stefanek of Little Rock, Arkansas, are using Excel to track their family budget to determine whether they can afford the monthly loan payments that would come with the purchase of a...
-
Look at Figure 29.28. a. Calculate the relative molecular mass of leucine enkephalin (C 28 H 37 N 5 O 7 ) using relative atomic masses. (A r values C = 12.0, H = 1.0, N = 14.0, O = 16.0) b. i. How is...
-
a. List the ions responsible for the M, [M + 2] and [M + 4] peaks in a mass spectrum of dibromomethane. b. What would be the mass-to-charge ratio and relative abundances of the major peaks with the...
-
Identify how each of the following separate transactions 1 through 10 affects financial statements. For increases, place a "+" and the dollar amount in the column or columns. For decreases, place a...
-
Crane Company was incorporated in Delaware in 2017. On November 2, 2024, the controller of the company entered into a forward contract to sell 36,000 British pounds for $1.5920 on March 1, 2025. The...
-
A neglectful hotel guest tosses an apple core off of a balcony with an initial horizontal velocity of 6.7 m/s. If the guest is 15.5 m high, how far horizontally will the apple core land from the...
-
What particular challenges do students face in online learning environments? Explain some preparation strategies to become a successful online student by detailing at least four steps to manage these...
-
The relationship between the Fahrenheit (F) and Celsius (C) scales is given by F = g(C) = C + 32. (a) Find g-1. g- (F) = What does g1 represent?
-
6. A soap bubble (n=1.33) having a wall thickness of 120 nm is floating in air. a. What is the wavelength of the visible light that is most strongly reflected? b. Explain how a bubble of different...
-
Explain the relationship between M&M's argument and the use of a residual dividend policy?
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
For each of the following pairs of compounds, identify the higher boiling compound and justify your choice: a. b.
-
Refer to Figure 1.10 and explain why (U/V) T is generally small for a real gas. Figure 1.10 Ideal gas Real gas Tranlaition rv-0 (4)A
-
Can a gas be liquefied through an isenthalpic expansion if J T = 0?
-
Problem 3 PCA Using the Fish Market Data Consider again the data in the previous problem, focusing just on the Perch case. Remember that you only use the test data for the final evaluation. The model...
-
A $24 credit to Revenue was posted as a $240 credit. By what amount is the Revenue account in error?
-
-/14 View Policies Current Attempt in Progress Blossom Enterprises relies heavily on a copier machine to process its paperwork. Recently the copy clerk has not been able to process all the necessary...

Study smarter with the SolutionInn App


