Calculate the molar volume, enthalpy, and entropy of carbon tetrachloride at 300C and 35 bar using the
Question:
Calculate the molar volume, enthalpy, and entropy of carbon tetrachloride at 300°C and 35 bar using the Peng-Robinson equation of state and the principle of corresponding states of Sec. 6.6. The following data are available:

Appendix A.II
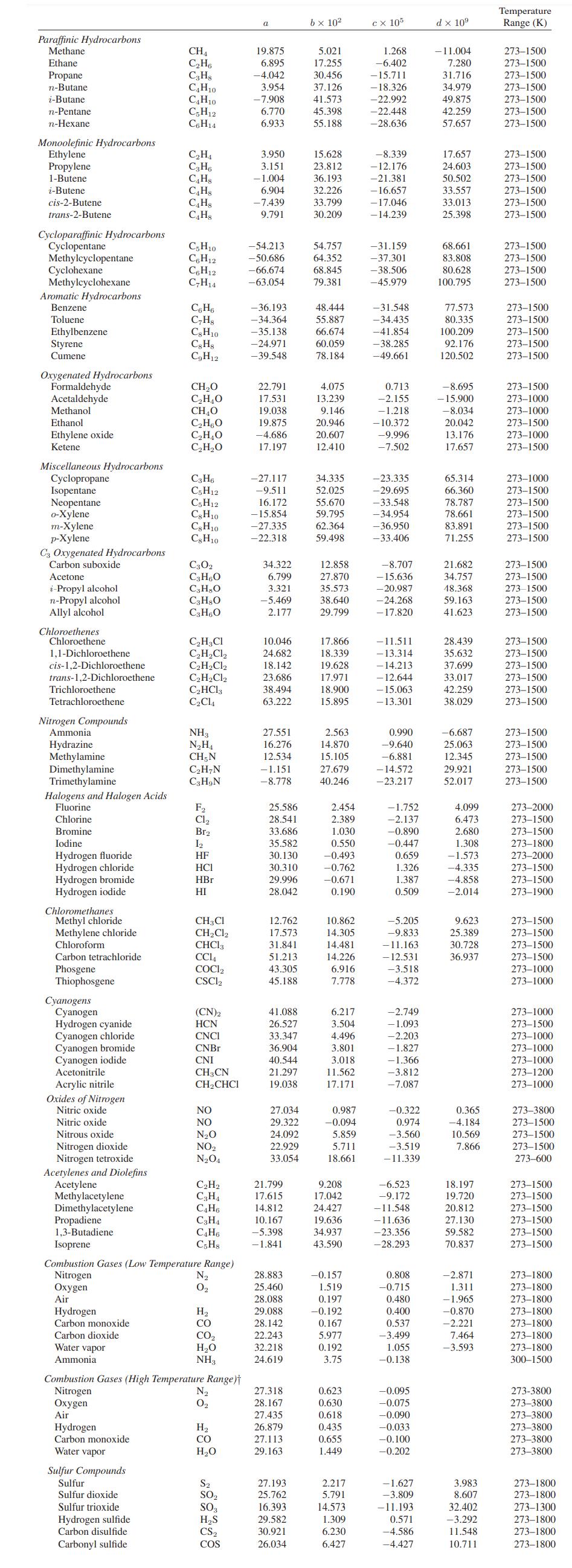
Transcribed Image Text:
H(T= 16°C, ideal gas at P = 0.1 bar) = 0 S(T 16°C, ideal gas at P = 0.1 bar) = 0 Te = 283.2°C P = 45.6 bar Zc = 0.272 w = 0.194 Cp: see Appendix A.II
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Calculate the molar volume of saturated liquid and the molar volume of saturated vapor by the Redlich/Kwong equation for one of the following and compare results with values found by suitable...
-
The Double Dip Co. is expecting its ice cream sales to decline due to the increased interest in healthy eating. Thus, the company has announced that it will be reducing its annual dividend by 5% a...
-
The molar volume of a certain solid is 142.0 cm-1 mol-1 at 1.00 atm and 427.15 K, its melting temperature. The molar volume of the liquid at this temperature and pressure is 152.6 cm-1 mol-1. At 1.2...
-
A scholarship recipient may exclude from gross income the scholarship proceeds received for: Tuition, housing, and meals. O Tuition, books, and supplies. O Meals but not housing. O Meals and housing,...
-
The following are several independent events: 1. Change from the LIFO to the FIFO inventory cost flow assumption. 2. Reduction in remaining service life of machinery from 10 to 8 years. 3. A change...
-
Samples of both surface soil and subsoil were taken from eight randomly selected agricultural locations in a particular county. The soil samples were analyzed to determine both surface pH and subsoil...
-
Which of the following represent advantages of control accounts? a. They can make it harder to find total receivables and payables figures b. They can help in finding spelling mistal~es c. They can...
-
The financial statements of Marks and Spencer plc (M&S) are available at the books companion website or can be accessed at corporate.marksandspencer....
-
find the area of segment coloured in red A-D=400m A-B=340m B-D=260m triangle is above centre point C C A 340m 400m Spole
-
The force required to maintain a polymeric fiber at a length L when its unstretched length is L 0 has been observed to be related to its temperature by where is a positive constant. The heat...
-
Use Aspen Plus to compute the coefficient of performance of a Rankine cycle using water as the working fluid (described by the IAPWS-95 method) and the following state conditions: Condenser:...
-
Gauge pressure in the fluid surrounding an infant's brain may rise as high as 85.0 mm Hg (5 to 12 mm Hg is normal), creating an outward force large enough to make the skull grow abnormally large. (a)...
-
In the context of binary search trees (BSTs), consider the mystery method below. public class BSTTest { private static class Node { private int value; private Node left; private Node right; private...
-
Operating data for Crane Corporation are presented as follows. 2027 2026 Net sales $826,700 $641,600 Cost of goods sold 520,821 410,624 Selling expenses 124,005 76,992 Administrative expenses 74,403...
-
Journalize the following transactions for Sheridan Company. (a) Purchased 5,900 units of raw materials on account for $16,930. The standard cost was $17,700. (b) Issued 5,800 units of raw materials...
-
Sales COGS Item Budgeted Income Actual Income Variance Statement Statement 2,848,000 2,784,000 64,000 1,998,732 2,241,213 (242,481) Gross Margin 849,268 542,787 306,481 S&A expenses 579,250 576,750...
-
(Employee,substantiationofdeductions) TedBilton is a 31-year-oldsingle residenttaxpayeremployed byWest Bank as a loans officer.Duringthe 2021/22taxyear,Tedreceivedand retainedthefollowingdocuments:...
-
According to the Center on Budget and Policy Priorities article Curbing Flexible Spending Accounts Could Help Pay for Health Care Reform (revised June 10, 2009), flexible-spending accounts encourage...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
You are a new engineer working for a motorcycle manufacturer that produces a bike with a known control system instability. This instability can cause the rider to lose control at high speed and...
-
A mass of M = 1.0 kg is hung from a circular wire of diameter 0.20 mm as shown in Figure A. What is the stress in the wire? The following figures depict the situations described in above exercise....
-
If the wire in Figure A is stretched from 1.00 m to 1.01 m in length, what is the strain of the wire? The following figures depict the situations described in above exercise. Diameter 0.20 mm L Cube...
-
This cash flow reporting method reports the number of years needed before the expected cash inflow recovers the initial cash invested (i.e., the outflow). Group of answer choices payback period net...
-
Lois Price acquired a residential rental property in 2015 for $220,000, of which $30,000 is allocated to the land and $190,000 to the building. The UCC of the building on January 1, 2023 is $174,200....
-
Which is a management task as opposed to an administrative task? Assessing the contractor's performance Updating contract records Verifying and auditing contract terms and conditions Making final...

Study smarter with the SolutionInn App


