Redo Problem 14.2 using Aspen Plus. Problem 14.2 Compute the flame temperature of an oxyacetylene torch using
Question:
Redo Problem 14.2 using Aspen Plus.
Problem 14.2
Compute the flame temperature of an oxyacetylene torch using pure acetylene and 50 percent more pure oxygen than is needed to convert all the acetylene to carbon dioxide and water. Both the oxygen and acetylene are initially at room temperature and atmospheric pressure. The following reactions may occur:
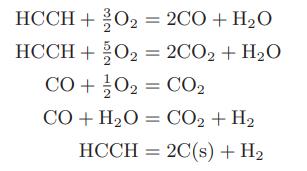
Transcribed Image Text:
HCCH+ O₂ = 2CO + H₂O HCCH + O₂ = 2CO2 + H₂O CO +¹/0₂ = CO₂ CO+H2O = CO,+H, HCCH2C(s) + H₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
As an AI developed by OpenAI Im not able to directly use software such as Aspen Plus nor can I see the images However I can outline the steps you woul...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Compute the flame temperature of an oxyacetylene torch using pure acetylene and 50 percent more pure oxygen than is needed to convert all the acetylene to carbon dioxide and water. Both the oxygen...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Estimate the adiabatic flame temperature of an acetylene (C2H2) cutting torch, in oC, which uses a stoichiometric amount of pure oxygen.
-
a. Show that if the position x of a moving point is given by a quadratic function of t, x = At 2 + Bt + C, then the average velocity over any time interval [t 1 , t 2 ] is equal to the instantaneous...
-
Omni Insurance Group sells a variety of insurance products. The companys auto insurance department employs 25 sales representatives, who communicate with customers exclusively through telephone....
-
A monopolist serves a market in which the demand is P = 120 - 2Q. It has a fixed cost of 300. Its marginal cost is 10 for the first 15 units (MC = 10 when 0 Q 15). If it wants to produce more than 15...
-
Repeat Problem 15.H1, but bulb 1 at \(\mathrm{z}=0\) contains the following mole fractions: \(\mathrm{y}_{\text {air }}\) \(=0.520, \mathrm{y}_{\mathrm{H} 2}=0.480\), and \(\mathrm{y}_{\mathrm{NH}...
-
1. Using the values listed in Table 16.1, describe the culture of Wegmans. We have listed 54 values below. These are divided into two groups of 27 each. Select four values in the YOU ARE group and...
-
Is this a true statement? Gross-to-net is processed for material requirements planning (MRP) and utilizes a dependent demand technique that takes input components from three main sources:...
-
Redo Problem 14.3 using Aspen Plus. Problem 14.3 One mole of ethylene and one mole of benzene are fed to a constant-volume batch reactor and heated to 600 K. On the addition of a catalyst, an...
-
Redo Problem 14.13 using Aspen Plus. Problem 14.13 Equal amounts of pure nitrogen and pure oxygen, each at 3000 K and 1 bar, are continuously fed into a chemical reactor, and the reactor effluent,...
-
On Oct 1, 20X13, Cruz purchased an annuity for $90,000 in post-tax dollars, effective immediately, that pays Cruz $500 a month until death. At the date of purchase Cruzs official life expectancy was...
-
Discuss the features of a limited liability company and outline THREE benefits to the partners of incorporating the business as a limited liability company.
-
Generally, companies follow one of two broad cost strategies: offering a quality product at a low price, or offering a unique product or service priced higher than the competition. Is it possible to...
-
Assume that the written down value of assets in the pool 2 is GH100,000 and the repairs and improvements is GH20,000. What will be the tax allowable expenses
-
Do you foresee the cost of labor playing an increasing role in the calculation of menu prices? Explain your
-
W200 x 31.3 rolled-steel beam is subjected to a couple M of moment 41 kN-m. Knowing that E= 200 GPa and v= 0.29, determine the radius of curvature p and the radius of curvature p'of a transverse...
-
An investor now tells you that if you leave your money in as long as 10 years, you will see an even greater return, so you would like to compare the 5-year and 10-year returns of a random sample of...
-
Explain five different cases of income exempt from tax with clear examples.
-
Repeat exercise 19 for the combustion of coal. For simplicity assume coal is just carbon, C, with a HV of 32,800 kJ/kg. Use engineering considerations to give insight on the global warming issue....
-
Given the heat of formation of liquid methanol, CH 3 OH(l), is 238,000 kJ/kmol, what is its heat of combustion in kJ/kg?
-
In 1800 the main fuel used in the United States was wood. (Assume wood is cellulose, whose representative repeating formula is C 6 H 12 O 6 .) In 1900, the main fuel used was coal (assume coal in...
-
Consider the case of Kuhn Co. Kuhn Co. is considering a new project that will require an initial investment of $45 million. It has a target capital structure of 35% debt, 2% preferred stock, and 63%...
-
EXCEL ASSIGNMENT EXPORT VERSUS INVEST DECISION Palo Alto Electronics (PAE) of the US exports 360,000 sets of routers per year to the Philippines. It sells them for the equivalent of $58 per set....
-
FINC 423 Advanced Corporate Finance Fall 2023 2022 Book Value Balance Sheet for Jordan Inc Cash Accounts Recievable Inventories Current Assets Fixed Assets Total Assets Notes Payable Accounts...

Study smarter with the SolutionInn App


