Repeat Problem 5.9 assuming that helium is described by the Peng-Robinson equation of state. Problem 5.9 High-pressure
Question:
Repeat Problem 5.9 assuming that helium is described by the Peng-Robinson equation of state.
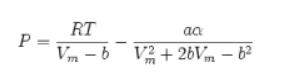
Problem 5.9
High-pressure helium is available from gas producers in 0.045-m3 cylinders at 400 bar and 298 K. Calculate the explosion equivalent of a tank of compressed helium in terms of kilograms of TNT. Assume helium is an ideal gas.
Transcribed Image Text:
P = RT aa Vm-b V2+2bVm – b²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To solve this problem we will need to use the PengRobinson equation of state to calculate the fugacity of helium at the given conditions The fugacity ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
High-pressure helium is available from gas producers in 0.045-m 3 cylinders at 400 bar and 298 K. Calculate the explosion equivalent of a tank of compressed helium in terms of kilograms of TNT....
-
In this problem, we consider the analysis of the combined information from both raters on the shoulder flexion (SF) scores in the posture measurement study. Thus, the questions below concern the data...
-
The molar heat capacity C P,m of SO 2 (g) is described by the following equation over the range 300 K < T < 1700 K: In this equation, T is the absolute temperature in kelvin. The ratios T/K ensure...
-
Suppose you make beautiful coffee tables out of juniper trees. Your materials cost for each table is $135. You pay your craftsman $25 per hour and it takes him 5 hours to complete one table. If you...
-
Several manufacturers introduced into the American market products known as all-terrain vehicles (ATVs). ATVs are motorized bikes that sit on three or four low-pressure balloon tires and are meant to...
-
Forty-five percent of all homes for sale in your local area sell quicklywithin 20 days of listing. Thirty-fve percent sell within 21 to 50 days. The remaining homes sell slowly, taking longer than 50...
-
Part 1. Gibbs, Gier, and Gill are partners and share income and loss in a 5:1:4 ratio. The partner ships capital balances are as follows: Gibbs, $303,000; Gier, $74,000; and Gill, $223,000. Gibbs de...
-
At the end of 2010, its first year of operations, the Swelland Company reported a pretax operating loss of $32,000 for both financial reporting and income tax purposes. At that time the company had...
-
find the percentage of direct materials added to whole units beginning WIp of 60,000 started and completed of 150,000 and ending work in process of 30,000
-
A laser pointer is kept at a constant and fixed height above the floor, but it can move horizontally back and forth in a straight line. A mirror is placed on a platform at a fixed distance from a...
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure of nitrogen as a function of specific volume at the two temperatures: a. 110 K b. 150 K
-
The Joule-Thomson coefficient, , given by is a function of temperature. The temperature at which = 0 is known as the inversion temperature. a. Use the van der Waals equation of state to determine...
-
The Air Travel Consumer Report, a monthly product of the Department of Transportations Office of Aviation Enforcement and Proceedings (OAEP), is designed to assist consumers with information on the...
-
Journalize the following transactions using the direct write-off method of accounting for uncollectible receivables. Apr. 1 Sold merchandise on account to Jim Dobbs, $7,200. The cost of the...
-
The four primary methods of gaining a competitive advantage are cost leadership, differentiation, defensive strategies and strategic alliances. Write detailed short notes on any two of these...
-
How does the concept of the circular economy propose a shift from traditional linear models of production and consumption, and what are the key strategies to achieve this transition at an industrial...
-
Explain the Product lifecycle in full detail and provide examples from the assigned company for each stage of the product lifecycle.
-
Boys get more attention in the classroom than girls. There's no doubt about it. Reams of studies show that teachers, from preschool to grade school, interact more with males than females. Especially...
-
Give the correct citation for the following case, include the parallel citations: Douglas D. Robberts versus Carroll E.
-
What is an access control list?
-
Use a plot over the range 0 x 5 to con rm that sin( ix) = i sinh x.
-
The function y(t) = 1 -! e -b t, where t is time and b > 0, describes many processes, such as the height of liquid in a tank as it is being filled and the temperature of an object being heated....
-
The following functions describe the oscillations in electric circuits and the vibrations of machines and structures. Plot these functions on the same plot. Because they are similar, decide how best...
-
What items should be reviewed to reduce ongoing practice cost? I. Supplies II. Phone/data plans III. Personal travel expenses IV. Building costs V. Malpractice insurance VI. Purchased services VII....
-
Which represents the correct order of deductions in the individual income tax formula? A. Gross income , Adjusted Gross income, Taxable income , credit, tax overpaid/ underpaid B. Gross income ,...
-
Jane provides a money purchase pension plan for her employees. She wants to fund the plan for each employee with either a term or whole life insurance policy. Which person does not pass the...

Study smarter with the SolutionInn App


