Consider the following compounds and formulas. (The formulas are written in such a way as to give
Question:
Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.)
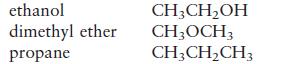
The boiling points of these compounds are (in no particular order) \(-42.1^{\circ} \mathrm{C},-23^{\circ} \mathrm{C}\), and \(78.5^{\circ} \mathrm{C}\). Match the boiling points to the correct compounds.
Transcribed Image Text:
ethanol dimethyl ether propane CH3CHOH CH3OCH3 CH3CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Matching Boiling Points to Compounds Heres the matching of boiling points to the correct compounds B...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Are there dangers with incarcerating young people in detention facilities? What about the potential for negative long-term impact? Using three documented examples of misconduct/mistreatment of youth...
-
A corporation has two divisions (the Eastern Division and the Western Division) that operate semiautonomously, with each developing and marketing its own products. However, to coordinate their...
-
c++. error: array must be initialized with a brace enclosed initializer main.cpp X 10 11 ii 12 13 14 4567 15 16 17 18 19 287288285 20 21 23 24 25 26 27 28 25285922223288 30 31 33 34 36 37 40 41 42 43...
-
A tank contains 1000 L of brine with 15 kg of dissolved salt. Pure water enters the tank at a rate of 10 L/min. The solution is kept thoroughly mixed and drains from the tank at the same rate. How...
-
a. For the Bayes net in Figure S13.37, we are given the query P(Z | + y). All variables are Boolean. Assume we run variable elimination to compute the answer to this query, with the following...
-
The following information relates to Ontario Components, Inc.: a. Calculate the company's return on common stockholders' equity for 2018 and 2019. b. Calculate the company's dividend yield for 2018...
-
The Project Data file provided for you contains information that you may use to help you complete certain questions. You will be directed to this file as required in the questions below. Canaans...
-
As with most bonds, consider a bond with a face value of $ 1 , 0 0 0 . The bond's maturity is 7 years, the coupon rate is 1 1 % paid annually, and the discount rate is 7 % What should be the...
-
Some of the physical properties of \(\mathrm{H}_{2} \mathrm{O}\) and \(\mathrm{D}_{2} \mathrm{O}\) are as follows: Account for the differences. (D is a symbol often used for \({ }^{2} \mathrm{H}\),...
-
For which molecule in each of the following pairs would you expect the stronger intermolecular forces? a. \(\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{NH}_{2}\) or \(\mathrm{H}_{2}...
-
What differences do you see between the U.S. version of American Idol and the version in other countries? Use material from this chapter to explain the reason for those differences.
-
When a net force Fo is applied to an object with mass Mo, it experiences an acceleration of 300 cm/s. What will be the acceleration of an object with mass 2-Mo when a net force of Fo/2 is applied to...
-
On July 1, an American auto dealer enters into a contract to purchase 20 Mercedes sports cars with payment to be made in Euros on November 1.Each car will cost Euros 40,000.Dealer want to hedge the...
-
Given the following account balances after closing entries are posted, what is the total credit amount on the Post-Closing Trial balance (assume normal account balances)? Cash Accounts Receivable...
-
A stock's current dividend is 6 and the required rate of return on this stock is 12%. If the dividend growth rate is 9%, what should be the current stock price? *
-
As part of its commitment to expanding its infrastructure, UNSW proposes to build a multistorey complex called the Science and Engineering Building which will provide teaching and learning spaces for...
-
Joe has achieved a position of some power in the institution in which he currently resides and works. In fact, things have gone so well that he has decided to divide the day- to- day operations of...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
Hydrogen peroxide, H 2 O 2 , is a nontoxic bleaching agent being used as a replacement for chlorine in industry and home laundries. The bleaching process is an oxidation, and when hydrogen peroxide...
-
Hydroxylamine, HONH 2 , is used to remove hair from animal hides and as a photoresist stripper in the electronics industry. Describe the structure of the hydroxylamine molecule in terms of hybrid...
-
Estimate the bond angles marked with arcs and lowercase letters in peroxyacetylnitrate, an eye irritant in smog: HC a C N=0 b 0-0 Peroxyacetylnitrate
-
ces Bond Returns and YTM You buy a(n) 9.8% coupon, 7-year maturity bond for $684.54. A year later, the bond price is $949.23. a. What is the new yield to maturity on the bond? (Do not round...
-
Required: 1. Show how you would handle the individual items in determining whether the company should continue to lease the space or convert it to a factory outlet. Use PV function in Excel, VDB...
-
Find the pdf of the sample median of a random sample of size four from a uniform(0,1) distribution and carefully sketch its graph. use the transformation: z1 = (Y2+Y3)/ 2 and Z2 = (Y3Y2)/ 2

Study smarter with the SolutionInn App


