The reaction [2 mathrm{NO}(g)+mathrm{Cl}_{2}(g) longrightarrow 2 mathrm{NOCl}(g)] was studied at (-10^{circ} mathrm{C}). The following results were obtained,
Question:
The reaction
\[2 \mathrm{NO}(g)+\mathrm{Cl}_{2}(g) \longrightarrow 2 \mathrm{NOCl}(g)\]
was studied at \(-10^{\circ} \mathrm{C}\). The following results were obtained, where
\[\text { Rate }=-\frac{d\left[\mathrm{Cl}_{2}ight]}{d t}\]
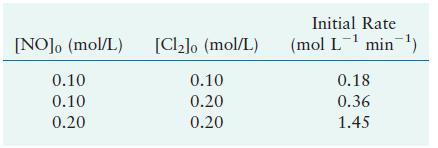
a. What is the rate law?
b. What is the value of the rate constant?
Transcribed Image Text:
[NO]o (mol/L) 0.10 0.10 0.20 [Cl]o (mol/L) 0.10 0.20 0.20 Initial Rate (mol L min) 0.18 0.36 1.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To determine the rate law for the given reaction we need to analyze how changes in the concentrations of the reactants affect the rate of the reaction The rate law can be written in the general form Rate k NOx Cl2y where k is the rate constant NO and Cl2 are the concentrations of the reactants and x and y are the orders of the reaction with respect to NO and Cl2 respectively We will use the provided initial rates and concentrations to deduce the values of x and y Lets examine how the initial rate changes when the concentration ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The enzyme carboxypeptidases catalyses the hydrolysis of polypeptides and here we consider its inhibition. The following results were obtained when the rate of the enzymolysis of...
-
The following results were obtained at 600 K for the de-composition of ethanol on an alumina (Al2O3) surface C2H5OH(g) C2H4(g) + H2O(g) a. Predict PTotal in torr at t = 80. s. b. What is the value of...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Given an array, Arr[] of integer numbers have size N. The task is to find out single or consecutive numbers from Arr[] with 0 sum. When found, print "1" otherwise "0". Example-1: Input: 4 (6,-2,3,-1)...
-
A direction field for a differential equation is shown. Draw, with a ruler, the graphs of the Euler approximations to the solution curve that passes through the origin. Use step sizes h = 1 and h =...
-
Indicate whether each of the following statements is true or false. A person of any age can make a will.
-
The following data were obtained prior to the acquisition of Gillette Company by Procter \& Gamble. Gillette Company, the Procter \& Gamble Company, and Colgate-Palmolive Company are three firms in...
-
The following data are for the two products produced by Shakti Company. The companys direct labor rate is $ 20 per direct labor hour (DLH). Additional information follows. Required 1. Compute the...
-
What was the result of the Christensens not complying with the 1031 exchange deadlines in their tax case? The exchange was postponed . They were granted a special exemption . Tax deferral was not...
-
The reaction \[2 \mathrm{I}^{-}(a q)+\mathrm{S}_{2} \mathrm{O}_{8}{ }^{2-}(a q) \longrightarrow \mathrm{I}_{2}(a q)+2 \mathrm{SO}_{4}{ }^{2-}(a q)\] was studied at \(25^{\circ} \mathrm{C}\). The...
-
The microwave spectrum of 1 H 35 Cl shows that the transition from J = 0 to J = 1 requires electromagnetic radiation with a wavelength of 4.85 10 4 m. Calculate the bond length of the 1 H 35 Cl...
-
Can the owner of a small business receive unemployment compensation? Explain.
-
Presented here are the comparative balance sheets of Hames Incorporated at December 31, 2023 and 2022. Sales for the year ended December 31, 2023, totaled $590,000. Check m HAMES INCORPORATED Balance...
-
A company is considering two mutually exclusive expansion plans. Plan A requires a $40 million expenditure on a large-scale integrated plant that would provide expected cash flows of $6.39 million...
-
How does the intricate interplay between organizational culture, stakeholder engagement, and strategic alignment serve as the cornerstone for efficacious Change Management initiatives within dynamic...
-
Cost Behavior Alisha Incorporated manufactures medical stents for use in heart bypass surgery. Based on past experience, Alisha has found that its total maintenance costs can be represented by the...
-
DLW Corporation acquired and placed in service the following assets during the year. Cost Basis $ 10,500 $ 21,000 $ 315,000 Asset Computer equipment Furniture Commercial building Assuming DLW does...
-
Product A is an end item and is made from two units of B and four of C. B is made of three units of D and two of E. C is made of two units of F and two of E. A has a lead time of one week. B, C, and...
-
Wimot Trucking Corporation uses the units-of-production depreciation method because units-of-production best measures wear and tear on the trucks. Consider these facts about one Mack truck in the...
-
Use the information in Fig. 2D.11 to estimate the length of (a) The CO bond in CO 2 ; (b) The CO and CN bonds in urea, OC(NH 2 ) 2 ; (c) The OCl bond in HClO; (d) The NCl bond in NOCl. CO B Bond...
-
Draw the Lewis structure and the VSEPR formula, list the shape, and predict the approximate bond angles of (a) PCl 3 F 2 ; (b) SnF4; (c) SnF 6 2- ; (d) IF 5 ; (e) XeO 4 .
-
Both NH 2 and NH 2 + are angular species, but the bond angle in NH 2 is less than that in NH 2 +. (a) What is the reason for this difference in bond angles? (b) Take the x -axis as lying...
-
DJI manufactures the Mavic Air 2 quadcopter and offered a 10/7/6 chain discount to primary customers. Drone Nerds is a primary customer and ordered 15 Mavic Air 2's for a total $19,500 list price. a....
-
just have a look if its correct qnd give me model ans This exercise stresses the relationships between the information recorded in a periodic inventory system and the basic elements of an Income...
-
Incluir materiales prcticos para potenciar el componente cinestsico/tctil de una leccin basada en la investigacin es un ejemplo de considerar: * a) La reflexin y la fijacin de objetivos b) Apelar a...

Study smarter with the SolutionInn App


