Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn 2+ to form the
Question:
Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn2+ to form the permanganate ion in an acidic solution.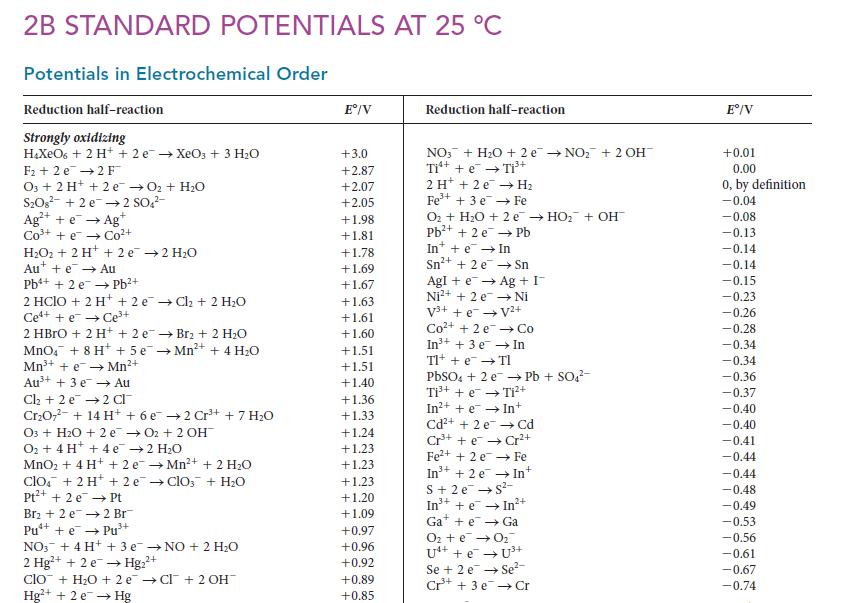
Transcribed Image Text:
2B STANDARD POTENTIALS AT 25 C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing HXeO6 + 2 H+ + 2 e XeO3 + 3 HO F+2 e 2 F O3 + 2 H+ + 2e O + HO SO8 +2e 2 SO4- Ag+ + e Ag C t HO + 2H+ + 2e 2 HO Aue Au Pb+ + 2 e Pb+ 2 HClO + 2 H+ + 2 eCl + 2 HO Ce++eCe+ 2 HBrO + 2H+ + 2 e Br2 + 2 HO MnO4 + 8 H+ + 5e Mn+ + 4HO Mn+ + eMn+ Au+ + 3 e Au Cl + 2 e 2 Cl CrO7- + 14 H+ + 6 e cot O3 + HO + 2 e O + 4H+ + 4e 2 HO MnO + 4H+ + 2e Mn+ + 2 HO clo + 2 H+ + 2 eClO3 + HO Pt+ + 2e Pt Br + 2 e Pu+ + ePu+ 2 Br 3+ 2 Cr+ + 7 HO O2 + 2 OH NO3 + 4H+ + 3 eNO + 2 HO 2 Hg+ + 2e Hg+ 2+ clo + HO +2e Cl + 2 OH Hg+ + 2e Hg E/V +3.0 +2.87 +2.07 +2.05 +1.98 +1.81 +1.78 +1.69 +1.67 +1.63 +1.61 +1.60 +1.51 +1.51 +1.40 +1.36 +1.33 +1.24 +1.23 +1.23 +1.23 +1.20 +1.09 +0.97 +0.96 +0.92 +0.89 +0.85 Reduction half-reaction NO3 + HO + 2e NO + 2 OH- Ti+ + e Ti+ 2 H + 2e H Fe+ + 3 e Fe O + HO + 2e HO + OH Pb+ + 2 e Pb In+ + e In Sn+ + 2 e Sn Ag + I Ni AgI + e Ni+ + 2 e V+ + e V+ Co+ + 2e In+ + 3 e Tl+ + e TI Co In PbSO4 + 2 ePb + SO4- Ti+ + e Ti+ In++eIn+ Cd+ + 2 e Cd Cr+ + eCr+ Fe+ + 2e Fe In +2 e S+ 2e S- + In +eIn2+ Gae Ga O + e 0 U4+ + e Se + 2 eSe- Cr+ + 3e Cr E/V +0.01 0.00 0, by definition -0.04 -0.08 -0.13 -0.14 -0.14 -0.15 -0.23 -0.26 -0.28 -0.34 -0.34 -0.36 -0.37 -0.40 -0.40 -0.41 -0.44 -0.44 -0.48 -0.49 -0.53 -0.56 -0.61 -0.67 -0.74
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
2 Clg will n...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use electrode potentials to answer the following questions, assuming standard conditions. (a) Do you expect permanganate ion (MnO4 ) to oxidize chloride ion to chlorine gas in acidic solution? (b)...
-
A customer made a reservation via telephone for a two-double bed room at the hotel. Unfortunately, once we arrived at the hotel, there was an error with the reservation and there were no rooms left...
-
(a) Use the data in Appendix 2B to decide which of ozone and fluorine is the stronger oxidizing agent in water. (b) Does youranswer depend on whether the reaction is carried out in acidic or basic...
-
The net income for Rips Curling Corp. for the year ended December 31, 2021, was $700,000. Rip had 50,000 ordinary shares outstanding at the beginning of the year. Rip declared and distributed a...
-
After genetically engineering a bacterium capable of breaking down crude oil, Ananda Chakrabarty sought to patent his creation under Title 35 U.S. Code Section 101, which states that "[w]hoever...
-
Refer to the data in Exercise 6-39. The president of Tiger Furnishings is confused about the differences in costs that result from using direct labor costs and machine-hours. Required a. Explain why...
-
Identify the five primary components of an accounting information system.
-
The following are the statements of financial position of Garden plc, its subsidiary Rose Ltd and its associate Petal Ltd: On 1 January 20X3 Garden plc acquired 75% of Rose Ltd for £300,000...
-
Marty is a principal who needs to contact the highest performing 15% of students in his school's current graduating class about an opportunity to join the school's academic honor society. At his...
-
Isoelectronic species have the same number of electrons. (a) Divide the following species into three isoelectronic groups: NH 3 , NO, NO 2 + , N 2 O, H 3 O + , O2 + . (b) Which species in each group...
-
Identify the products and write a balanced equation for the reaction of hydrogen with (a) Chlorine; (b) Sodium; (c) Phosphorus; (d) Copper.
-
The rigid L-shaped member ABF is supported by a ball-and-socket joint at A and by three cables. For the loading shown, determine the tension in each cable and the reaction at A.
-
Red Horse, Inc. a small beer brewery. The beer is processed in a single brewery into light beer, dark beer, and an impure byproduct. There were no beginning inventories or work-in-process on...
-
Pikachu Co. had 4,000 units work in process at November 1, 2017, which were 50% complete to conversion cost. During November, 27,000 units was completed. At November 30, 3,000 units remained in WIP...
-
A credit card company has a detection service that alerts the user if an unusually high charge is placed on a credit card. Given the following list of charges for one client 110 189 89 128 154 127...
-
The Papa Manufacturing Company uses the perpetual inventory system in controlling its materials.Data relating to Material X-5 during March 2018 are given below: March 1:Balance, 1,000 units at P125...
-
You are given the following data on the manufacturing operations of the entity for August, 2020: In process, August 1 (8,000 units, 25% done) Factory costs: Material cost P 2.100 21,000 Labor cost...
-
What are the differences of the following: required rate of return on a stock, expected rate of return on a stock, actual or realized rate of return on a stock.
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
The shear stressstrain diagram for a solid 50-mm-diameter shaft can be approximated as shown in the figure. Determine the torque required to cause a maximum shear stress in the shaft of 125 MPa. If...
-
A steel alloy core is bonded firmly to the copper alloy tube to form the shaft shown. If the materials have the Ï-γ diagrams shown, determine the torque resisted by the core and the...
-
The shaft consists of two sections that are rigidly connected. If the material is elastic perfectly plastic as shown, determine the largest torque T that can be applied to the shaft. Also, draw the...
-
We have learned several business analytic topics in this course. Discuss how you might use them in your personal life or your job, such as managing your grocery purchasing, automobile maintenance,...
-
Problem 24-4A (Algo) Applying net present value and profitability index LO P3 Rowan Company is considering two alternative investment projects. Each requires a $268,000 initial investment. Project A...
-
Which of the following statements is false? A. A C corporation with taxable income of $100,000 in the current year will have a tax liability of $21,000. B. A corporation cannot deduct net capital...

Study smarter with the SolutionInn App


