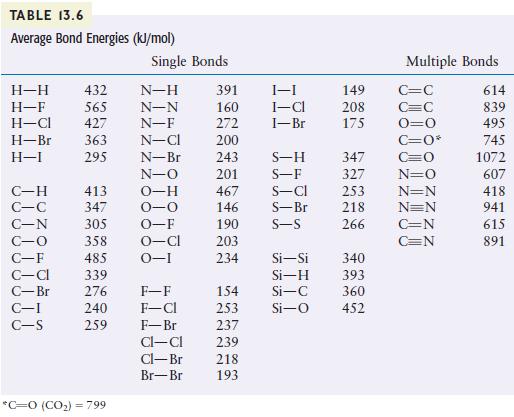
Using the bond energies listed in Table 13.6, calculate H for the reaction of methane with chlorine
Question:
Using the bond energies listed in Table 13.6, calculate ΔH for the reaction of methane with chlorine and fluorine to give Freon-12 (CF2Cl2).
![]()
Transcribed Image Text:
CH(g) + 2Cl(g) + 2F(g) CFCl(g) + 2HF(g) + 2HCl(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The idea here is to break the bonds in the reactants to give indi vidual atoms and then assemble the...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Q3. Modulus of rupture of concrete is a measure of a) flexural tensile strength b) direct tensile strength c) compressive strength d) split tensile strength Q4. For a longitudinal reinforcing bar in...
-
(a) For what nonzero values of does the function y = sin kt satisfy the differential equation yn + 9y = 0? (b) For those values of k, verify that every member of the family of functions y = A sin kt...
-
In Exercises the rectangular coordinates of a point are given. Plot the point and find two sets of polar coordinates for the point for 0 0 < 2. (-3, 4)
-
The following production information about quality costs has been gathered for September: Using these data, calculate the following: a. Total cost to rework b. Profit lost from not reworking all...
-
Ann took Bonnies watch before Bonnie was aware of the theft. Bonnie discovered her loss immediately and pursued Ann. Ann pointed a loaded pistol at Bonnie, who, in fear of being shot, allowed Ann to...
-
Consider four different stocks, all of which have a required return of 18.5 percent and a most recent dividend of $3.65 per share. Stocks W, X, and Y are expected to maintain constant growth rates in...
-
Give the Lewis structure for each of the following. a. HF b. N 2 c. NH 3 d. CH 4 e. CF 4 f. NO +
-
Choose the largest ion in each of the following groups. a. Li + , Na + , K + , Rb + , Cs + b. Ba 2+ , Cs + , I , Te 2
-
Identifying Increase and Decrease Effects on Balance Sheet Elements Complete the following table by entering either the word increases or decreases in eachcolumn. Debit Credit Assets Liabilities...
-
For this assessment you will read the SWOT analysis for Netflix then addresses the following: Provide two qualitative data sources that would provide information that would assist Netflix in making...
-
The project team has started the risk management plan and needs now to identify, assess, and manage the project risks. In order to identify individual project risks, the team will examine those tasks...
-
Simon Company's year-end balance sheets follow. At December 31 Assets Current Year 1 Year Ago 2 Years Ago Cash Accounts receivable, net $ 32,368 93,813 $ 38,971 68,198 $ 38,230 53,075 Merchandise...
-
When a consumer finds a way to claim a bigger prize than the company likely intended, what happens? What is the company legally obligated to do, and what does it actually do? Are the promotions...
-
1. -What are perceptions? - How can perceptions affect customer relations? Please provide at least 2 examples, preferably from your own business experiences. 2. Situation: You often hear one of your...
-
DAT, Inc., produces digital audiotapes to be used in the consumer audio division. DAT lacks sufficient personnel in its inventory supply section to closely control each item stocked, so it has asked...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Identify the hybrid orbitals used by the atom in boldface red type in each of the following molecules: (a) H 2 C C CH 2 ; (b) H 3 C C H 3 ; (c) CH 3 N N N; (d) CH 3 C O OH.
-
(a) Draw the molecular orbital energy-level diagram for N 2 and label the energy levels according to the type of orbitals from which they are made, whether they are - or -orbitals, and whether they...
-
Draw the Lewis structure for each of the following molecules or ions and give the number of electrons about the central atom: (a) SF 6 ; (b) XeF 2 ; (c) AsF 6 ; (d) TeCl 4 .
-
A sample of 18 items provides a sample standard deviation of 4.9 . Test the following hypotheses using a=0.05. What is your conclusion? Use both the p-value approach and the critical value approach....
-
Hello tutors! Please help me with this task. I will upvote those who will. Please provide a step-by-step solution to each problem. It must be presented with the graphic organizer table chart. Follow...
-
When an employer does not have a sophisticated time-keeping system, it is valid to accrue a percentage of wages at end-of-period based on: a. it is never valid to estimate an end-of-period...

Study smarter with the SolutionInn App


