With the help of Table 9C.1, determine the coordination number of the metal ion in each of
Question:
With the help of Table 9C.1, determine the coordination number of the metal ion in each of the following complexes:
(a) [NiCl4]2–;
(b) [Ag(NH3)2]+;
(c) [PtCl2(en)2]2+;
(d) [Cr(edta)]–.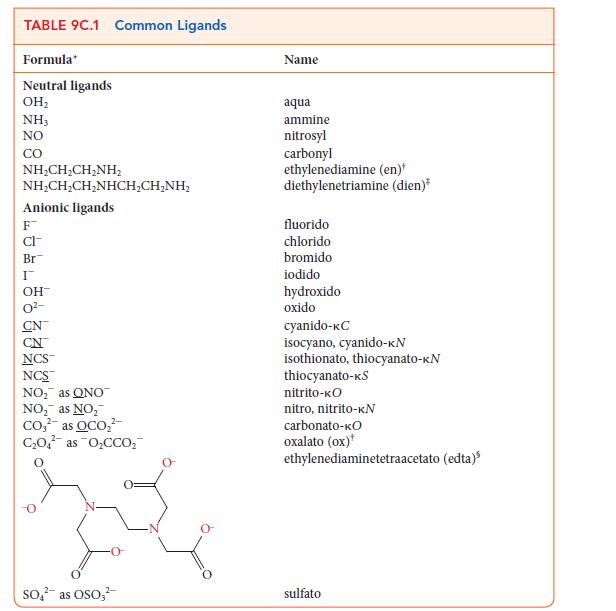
Transcribed Image Text:
TABLE 9C.1 Common Ligands Formula Neutral ligands OH NH3 NO CO NHCH,CH_NH, NHCH,CH,NHCH,CHNH, Anionic ligands F CI- Br I OH- CN NCS NCS NO as ONO NO as NO Co as OCO C0 as OCCO 2- SO as OSO3 Name aqua ammine nitrosyl carbonyl ethylenediamine (en) diethylenetriamine (dien)* fluorido chlorido bromido iodido hydroxido oxido cyanido-KC isocyano, cyanido-KN isothionato, thiocyanato-KN thiocyanato-KS nitrito-KO nitro, nitrito-KN carbonato-KO oxalato (ox) ethylenediaminetetraacetato (edta) sulfato
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 4 b 2 c ...View the full answer

Answered By

Ayush Mishra
I am a certified online tutor, with more than 3 years of experience in online tutoring. My tutoring subjects include: Physics, Mathematics and Mechanical engineering. I have also been awarded as best tutor for year 2019 in my previous organisation. Being a Mechanical Engineer, I love to tell the application of the concepts of science and mathematics in the real world. This help students to develop interest and makes learning fun and easy. This in turn, automatically improves their grades in the subject. I teach students to get prepared for college entry level exam. I also use to teach undergraduate students and guide them through their career aim.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
IN DETAILED - Pseudo code for findLCS() appears after the pseudo code for cutRod(). int cutRod( int [] p ) Initialize. Set n to the length of the prices array p. Allocate space for an array r from 0...
-
The Company XYZ has 1173 blocks of building for its business operation, where each block has 7 floors. The distance between each floor is 7 meters. ] (ii) Give a function run2diff which can be...
-
With the help of Table 9C.1, determine the coordination number of the metal ion in each of the following complexes: (a) PtBr 2 (NH 3 ) 2 ; (b) [Ni(en) 2 I 2 ] + ; (c) [Co(ox) 3 ] 3 ; (d) [Mn(CO) 5 ] ...
-
This is a stocklist case arising under 220(b) of our [Delaware] General Corporation Law. The issue is whether a shareholder states a proper purpose for inspection under our statute in seeking to...
-
An Australian company sought to sell sparkling wine in New Zealand. The wine was made in Australia from grapes grown in Australia, but was packaged in bottles that included the word "champagne" on...
-
Consider the Business Application item, referred to in Question 4-19. Suppose the company had signed a lease for five years and had two years remaining on the contract. Would the remaining lease...
-
When special journals are used, where are cash payments by check recorded?
-
Hummer Company purchased a delivery truck. The total cash payment was $30,020, including the following items. Negotiated purchase price..........................$24,000 Installation of special...
-
Use the (x,y) coordinates in the figure to find the value of the trigonometric function at the indicated real number, s, or state that the expression is undefined. 7x sin 6 K2 V K 12 42 (44) (4)...
-
Suppose you are working in a laboratory synthesizing complexes of Mn and Fe. You have created a new ligand and need to know how strong- or weak-field ligands might affect the properties of your new...
-
One method used to produce hydrogen as a fuel decomposes methanol in the reaction CH 3 OH(l) 2 H 2 (g)+ CO(g). (a) What is the standard reaction enthalpy of this process? (b) What is the standard...
-
What are the major elements of the cost of capital?
-
A patient has developed a fluid imbalance secondary to MDMA use which has resulted in a condition called Syndrome of Inappropriate Antidiuretic Hormone Secretion (SIADH) 1) On A concept map, start at...
-
In an epicyclic gear train, as shown in Fig.15.25, the number of teeth on wheels A, B, and C are 50, 25, and 52 respectively. If the arm rotates at 420 rpm cw, find (a) speed of wheel C when A is...
-
Why does flexibility in NoSql make it easier? What is an example.
-
1. TRUE/FALSE questions about electricity markets a. During off-peak hours, incentives to exercise market power diminish. (TRUE/FALSE) b. In a competitive electricity market, higher natural gas...
-
Give any four advantages of Cost Accounting. ii. What is meant by Safety Stock? iii. What is Flux Rate of Labour Turnover? How is it determined? iv. What do you mean by "Economic Batch Quantity" in...
-
A firm has debt of $8,000, a leveraged value of $18,800, a cost of debt of 8.75 percent, a cost of equity of 13 percent, and a tax rate of 35 percent. What is the firm's weighted average cost of...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
If the shaft is subjected to a uniform distributed torque of t = 20 kN m/m, determine the maximum shear stress developed in the shaft. The shaft is made of 2014-T6 aluminum alloy and is fixed at A...
-
The 6061-T6 aluminum bar has a square cross section of 25 mm by 25 mm. If it is 2 m long, determine the maximum shear stress in the bar and the rotation of one end relative to the other end. 1.5 m 20...
-
Segments AB and BC of the assembly are made from 6061-T6 aluminum and A992 steel, respectively. If the allowable shear stress for the aluminum is (Ï allow ) al = 12 ksi and for the steel (Ï...
-
Data Analytics at HydroHappy Excel DA9.1 Data visualization can be used to analyze trends in production levels. HydroHappy has developed a new marketing plan that looks very promising for increased...
-
Doug Davidson, majority stockholder and president of Davidson, Inc., is working with his top managers on future plans for the company. As the company's managerial accountant, you've been asked to...
-
Question 3 6 pts Miliea Industries uses a perpetual inventory system. On May 1, beginning inventory consists of 10 items at a cost of $10 each. On May 3, 10 items are purchased at $12 each. On May 8,...

Study smarter with the SolutionInn App


