Estimate the LOC for (a) carbon monoxide and (b) heptane using Equation 6-15. Compare to experimental values
Question:
Estimate the LOC for
(a) carbon monoxide and
(b) heptane using Equation 6-15. Compare to experimental values in Table 6-3.
Table 6-3:
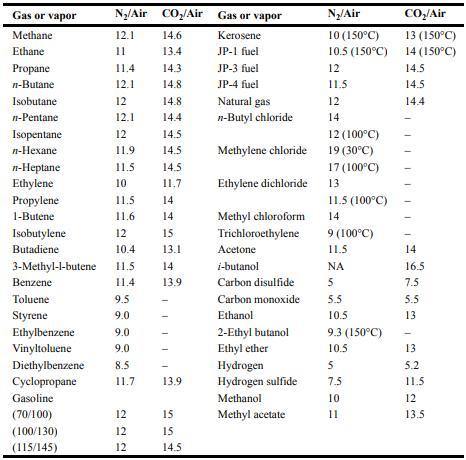
Equation 6-15:
![]()
Transcribed Image Text:
Gas or vapor Methane Ethane Propane n-Butane Isobutane n-Pentane Isopentane n-Hexane n-Heptane Ethylene Propylene 1-Butene Isobutylene Butadiene Benzene Toluene Styrene Ethylbenzene Vinyltoluene 12 11.9 11.5 10 11.5 11.6 12 15 10.4 13.1 3-Methyl-1-butene 11.5 14 11.4 13.9 Diethylbenzene Cyclopropane Gasoline (70/100) N/Air CO/Air Gas or vapor Kerosene (100/130) (115/145) 12.1 11 11.4 12.1 12 12.1 9.5 9.0 9.0 9.0 8.5 11.7 222 12 14.6 13.4 12 12 14.3 14.8 14.8 14.4 14.5 14.5 14.5 11.7 14 14 1 13.9 15 15 14.5 JP-1 fuel JP-3 fuel JP-4 fuel Natural gas n-Butyl chloride Methylene chloride Ethylene dichloride Methyl chloroform Trichloroethylene Acetone i-butanol Carbon disulfide Carbon monoxide Ethanol 2-Ethyl butanol Ethyl ether Hydrogen Hydrogen sulfide Methanol Methyl acetate N/Air 10 (150C) 10.5 (150C) 12 11.5 12 14 12 (100C) 19 (30C) 17 (100C) 13 11.5 (100C) 14 9 (100C) 11.5 5. 5.5 10.5 9.3 (150C) 10.5 5 7.5 10 11 COp/Air 13 (150C) 14 (150C) 14.5 14.5 14.4 1 14 16.5 7.5 5.5 13 13 5.2 11.5 12 13.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Equation 615 uses the Lower Flammable Limit LFL to compute the Lower Oxygen Content LOC The LFL is t...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
A helicopter spots two landing pads below. The straight-line distance from the helicopter to the pads is 14 miles to the landing Pad A and 8 miles to landing Pad B. If the landing pads are 20 miles...
-
Provide a link to the artifact you plan to analyze for your Rhetorical Analysis. In roughly, 300 words do the following: Summarize the artifact + Explain why you chose the artifact Discuss the...
-
Estimate the upper and lower flammable limits for carbon monoxide and heptane using the stoichiometric method via Equations 6-10 and 6-11 in the text. Compare to experimental values provided in...
-
machine element is made of an alloy with Yield strength of 200 Mpa and fracture strain is 0.08. A machine element is characterized by the normal stress components as shown in the figure. The Answer...
-
Verify that the expressions for the volumes of the first four shapes in Fig. 5.21 on page 261 are correct.
-
Which of the following is true about Social Security and Medicare taxes, as they pertain to earnings limits? (Select all that apply) A. Social Security tax applies to all earnings, regardless of...
-
Humans collectively exhale approximately \(6.4235 \times 10^{12}\) pounds of carbon dioxide per year. There are approximately \(7.647 \times 10^{9}\) humans currently living on Earth. How many pounds...
-
Jeff Bowman and Kristi Emery, who have ending capital balances of $100,000 and $60,000, respectively, agree to admit two new partners to their business on August 18, 20--. Dan Bridges will buy...
-
Use the following information for the next three questions: Vishal Noronha needs to prepare a valuation for Sindhuh Enterprises. Noronha has collected the following information for his analysis. It...
-
Draw an approximate flammability triangle diagram for methyl alcohol. Use published flammability data from Appendix B and Table 6-3. If a gas containing 20\% methyl alcohol, \(5 \%\) oxygen, and \(75...
-
Estimate the LOC of ethylene using Equations 6-15 and 6-16 in the textbook. Compare to the experimental value in Table 6-3. Table 6-3: Equation 6-15: Equation 6-16: Gas or vapor Methane Ethane...
-
What are ethics and how do ethics apply to business?
-
Because existing staff are fully scheduled, if the firm is awarded the contract, it must hire one new staff member at a salary of $52,000 to handle the additional workload. The managing partner is...
-
During March, Hart Company incurred the following costs on Job 109 for the manufacture of 200 motors: Original cost accumulation: Direct materials Direct labor Factory overhead (150% of DL) Direct...
-
An asset originally purchased for $12,122,100 has an estimated salvage value of $7.622.100 after five years of use Using the Sum-of- Year's-Digits (SOYD) method of book depreciation (a) The Book...
-
In this article, answer the question. Why are great networking opportunities most important? Explain why you feel this way....
-
Brighton Services repairs locomotive engines. It employs 100 full-time workers at $16 per hour. Despite operating at capacity, last year's performance was a great disappointment to the managers. In...
-
On December 1, 2011, Barnum Company (a U.S.-based company) entered into a three-month forward contract to purchase 1,000,000 ringgits on March 1, 2012. The following U.S. dollar per ringgit exchange...
-
If someone's Z-score for a variable was 0.67. Their score is a significant extreme score. Their score is not significant. O Their score is slightly above average. O Their score is an outlier.
-
A robotic-wheeled vehicle that contains science instruments is used to study the geology of Mars. The rover weighs 408 lb on Earth. (a) In the USCS dimensions of slugs and lbm, what is the rovers...
-
Calculate various fuel quantities for Flight 143. The plane already had 7682 L of fuel on board prior to the flight, and the tanks were to be filled so that a total of 22,300 kg were present at...
-
Printed on the side of a tire on an all-wheel-drive sport utility wagon is the warning Do not inflate above 44 psi, where psi is the abbreviation for the pressure unit pounds per square inch (lb/in 2...
-
. Save & Exit Subm 2 Whitman has a direct labor standard of 2 hours per unit of output. Each employee has a standard wage rate of $24.50 per hour. During July, Whitman paid $196,000 to employees...
-
. Help Save & E 25 Regent Corp. uses a standard cost system to account for the costs of its one product. Materials standards are 3 pounds of material at $14 per pound, and labor standards are 4 ...
-
What does the mystery1 method do in terms of h? The class Node is a regular BST node with key, value, left, and right subtrees.. public Nodeck, V> mystery1 (Node h) { if (h. left nu LU)...

Study smarter with the SolutionInn App


