Dilute aqueous ethanol (about 2-3%) is oxidized to acetic acid by the action of pure oxygen at
Question:
Dilute aqueous ethanol (about 2-3%) is oxidized to acetic acid by the action of pure oxygen at 10 atm in a trickle bed reactor packed with palladium-alumina catalyst pellets and kept at 30°C. According to Sato et al., Proc. First Pacific Chem. Eng. Congress, Kyoto, p. 197,1972, the reaction proceeds as follows: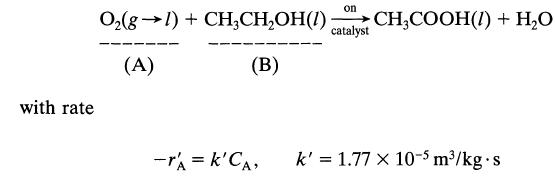
Find the fractional conversion of ethanol to acetic acid if gas and liquid are fed to the top of a reactor in the following system:

Transcribed Image Text:
with rate on catalyst O₂(g) + CH₂CH₂OH(1) CH3COOH(1) + H₂O (A) (B) -rA = k'CA, k' = 1.77 x 10-5 m³/kg-s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The material balance equations for the gas and liquid phases of a trickling bed reactor will be used to tackle this issue The kinetics of the reaction ...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Instead of using a trickle bed reactor for ethanol oxidation (see previous problem), let us consider using a slurry reactor. For this type of unit Take all flows and other values from the previous...
-
Ethyl alcohol can be bacterially oxidized to acetic acid in the following two-step fermentation sequence: 2 C2H5OH + O2 2 CH3CHO + 2 H2O 2 CH3CHO + O2 2 CH3COOH If the alcohol-containing feedstock...
-
Trickle bed reactors is widely used in the oil industry because of the advantages offered. In a trickle bed reactor, the oxidation of ethanol is to be carried out. The reaction is first order with...
-
In a survey of 1,002 people, 701 (or 70%) said that they voted in the last presidential election (based on data from ICR Research Group). The margin of error was 3 percentage points. However, actual...
-
What is marketing and what is its primary goal?
-
Calculate the turbine specific speed of the turbine in Prob. 1486E using customary U.S. units. Is it in the normal range for a Francis turbine? If not, what type of turbine would be more appropriate?...
-
Generally accepted accounting principles are: a. the guidelines used to resolve ethical dilemmas. b. established by the Internal Revenue Service. c. primarily established by the Financial Accounting...
-
As in Question 7.3 of Chapter 14, Samanthas utility function is U(Y) = Y 0.5 . She tries to maximize her expected utility. She owns a car for business that she will have to replace if it is stolen....
-
Finch Company, which expects to start operations on January 1 , year 2 , will sell digital cameras in shopping malls. Finch has budgeted sales as indicated in the following table. The company expects...
-
FCC reactors are among the largest processing units used in the petroleum industry. Figure P1.3 shows an example of such units. A typical unit is 4-10 m ID and 10-20 m high and contains about 50 tons...
-
Predict the conversion of glucose to sorbitol in a stirred slurry reactor using pure hydrogen gas at 200 atm and 150C. The catalyst used is porous Raney nickel, and under these conditions Brahme and...
-
The city of Lydell expended federal awards from the following programs during 2019. Assume the auditor has given an unqualified opinion on the financial statements and reports no material weaknesses...
-
Identify four private sector organize ions associated with the public accounting profession and briefly desciibe their principal activities.
-
Becky Maupin, CPA, has been asked to compile the financial statements of George's Diner, a nonpublic company, for the year ended April 30, 19X5. a. What understanding should Maupin establish with...
-
In a small group, have each individual take on the role as spokesperson for the American Federation of State County and Municipal Employees, the Service Employees International Union, and the America...
-
As a condition for a public offering of securities by your largest and most important audit client, the underwriter requires a comfort letter containing the following items. 1. An unequivocal...
-
John Alvis, CPA, has completed his review of the interim financial information of Wilson and Love, a public company, for the six months ended June 30, 19X5. a. Discuss the form of report that Alvis...
-
Let S = $100, K = $105, r = 8%, T = 0.5, and = 0. Let u = 1.3, d = 0.8, and n = 1. a. What are the premium, , and B for a European call? b. What are the premium, , and B for a European put?
-
Identify the tax issues or problems suggested by the following situations. State each issue as a question. Jennifer did not file a tax return for 2007 because she honestly believed that no tax was...
-
A CSTR is being operated at steady state. The cell growth follows the Monod growth law without inhibition. The exiting substrate and cell concentrations are measured as a function of the volumetric...
-
Diabetes is a global epidemic affecting more than 240 million people worldwide. The vast majority of the cases are Type 2. Recently a drug, Januvia (J), was discovered to treat Type 2 diabetes. When...
-
In the summer of 2009, ExxonMobil decided to invest 600 million dollars on developing algae as an alternative fuel and their TV commercials on this initiative were recently shown (e.g., December...
-
ACC - 2 4 0 Gradebook Question 6 - Topic 7 Assignment - dots Topic 7 Assignment i Help Save & Exit Submit 6 7 . 5 points eBook References Lakeside Incorporated produces a product that currently sells...
-
Jasper Company has 6 3 % of its sales on credit and 3 7 % for cash. All credit sales are collected in full in the first month following the sale. The company budgets sales of $ 5 3 2 , 0 0 0 for...
-
Playtown Corporation purchased 7 5 percent of Sandbox Company common stock and 4 0 percent of its preferred stock on January 1 , 2 0 6 , for $ 2 7 0 , 0 0 0 and $ 8 0 , 0 0 0 , respectively. At the...

Study smarter with the SolutionInn App


