Find the conversion after 1 hour in a batch reactor for A R, -A= 3C05 mol liter.hr'
Question:
Find the conversion after 1 hour in a batch reactor for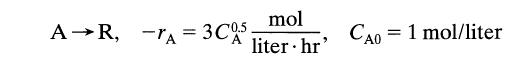
Transcribed Image Text:
A R, -A= 3C05 mol liter.hr' CA0 = 1 mol/liter
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To find the conversion after 1 hour in a batch reactor for the given reaction we need to integrate the rate law to find the relation between concentration and time The provided image gives the reactio...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
For the reaction A R, second-order kinetics and C A0 = 1 mol/liter, we get 50% conversion after 1 hour in a batch reactor. What will be the conversion and concentration of A after 1 hour if C A0 =...
-
What is it like to be a minority within the majority culture? Here are some diversity-related scenarios that you, as a manager, might tackle. Diversity is a very touchy subject, so I hope our...
-
HealthySight is a manufacturer of high-quality lenses for sunglasses and ski goggles. HealthySight uses a standard process costing system and carries inventories at standard. In May 2001, the...
-
Fill in the blanks in the chart below: Year Years since 1960 1960 1965 1970 1975 1980 1985 1990 1995 1996 1997 1998 1999 2000 2001 2003 2005 2006 2007 2008 2009 2010 2011 0 5 [a] [b] [c] [d] [e] [f]...
-
Consider a put contract on a T-bond with an exercise price of 101 12/32. The contract represents $100,000 of bond principal and has a premium of $750. The actual T-bond price is currently 100 1/32....
-
Harvey is a self-employed accountant with earned income from the business of $120,000 (after the deduction for one-half of his self-employment tax). He uses a defined contribution Keogh plan. What is...
-
Microsoft, one of the most successful businesses in the history of commerce, has accumulated a large investment portfolio, which in a recent year consisted of the following (in millions): In...
-
The following data relating to direct materials cost for October of the current year are taken from the records of Good Clean Fun Inc., a manufacturer of organic toys: Quantity of direct materials...
-
There is a % column right beside each balance sheet account for each year. Go back and look at the template provided. The text explains the difference between a balance sheet and a common - size...
-
A small reaction bomb fitted with a sensitive pressure-measuring device is flushed out and then filled with pure reactant A at 1-atm pressure. The operation is carried out at 25OC, a temperature low...
-
Enzyme E catalyzes the transformation of reactant A to product R as follows: If we introduce enzyme (C E0 = 0.001 mol/liter) and reactant (C A0 = 10 mol/liter) into a batch reactor and let the...
-
Give three examples of the uses of an accounting system.
-
Minker expresses an unqualified opinion concerning the financial statements of the Zee Company, in which statements the basis of valuation of the fixed assets is not shown. She is aware of the fact...
-
Consider two ways of looking at the profit of a business: an increase in the book wealth of the company; and the net result of the companys trading operations (revenue less expenses). What do these...
-
Outline an income statement showing each of the main category headings using the business functions format.
-
A friend of yours owns a shop selling CDs and posters for the 12- to 14-year-old market. From the following information advise him on the potential problems that may be encountered in the valuation...
-
Tartantrips Ltd, a company in Scotland, operates several ferries and has a policy of holding several in reserve, due to the weather patterns and conditions of various contracts with local...
-
Consider the following two bonds which make semiannual coupon payments: a 20 year bond with a 6% coupon and 20% yield, and a 30-year bond with a 6% coupon and a 20% yield. a. For each bond, compute...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
An RTD experiment was carried out in a nonideal reactor that gave the following results: a. What are the mean residence time, tm, and variance 2 ? b. What is the fraction of the fluid that spends a...
-
Go to Extra Help then click on Videos of Tips on Studying and Learning. Go to Chapter 1 (http://www.umich.edu/~elements/6e/01chap/obj.html#/videotips/). 1. View one of the 5- to 6-minute video...
-
Go to Chapter 1 Extra Help on the Web site and click on LearnChemE ScreenCasts (http://www.umich.edu/~elements/6e/01chap/learn-chemevideos.html) the How to Study screencast and list three ways that...
-
Healthy Life Co . is an HMO for businesses in the Fresno area. The following account balances appear on Healthy Life s balance sheet: Common stock ( 3 , 0 0 0 , 0 0 0 shares authorized; 2 , 2 0 0 , 0...
-
Try to answer the following questions: 1 . Brown Corp purchased an equipment at $ 4 0 , 0 0 0 . This equipment can be used for 1 0 years. At the end of its useful life, Brown estimates it will worth...
-
1 Required information [ The following information applies to the questions displayed below. ] Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the...

Study smarter with the SolutionInn App


