For a gas reaction at 400 K the rate is reported as (a) What are the units
Question:
For a gas reaction at 400 K the rate is reported as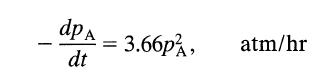
(a) What are the units of the rate constant?
(b) What is the value of the rate constant for this reaction if the rate equation is expressed as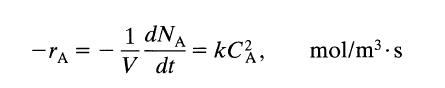
Transcribed Image Text:
dp A dt 3.66p, atm/hr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Hence the value of rate constant is 12013 with units lmolh A gas phase reaction ...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
What are component units of a government, and how are they reported on the government-wide financial statements?
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
What are equivalent units of production?
-
Review Questions: 1. What is the theory on which Rockwell hardness testing is based? 2. What is the purpose of the minor load in Rockwell hardness testing? 3. What are the advantages of the Rockwell...
-
1. Explore the Web sites of other companies to learn how they get feedback from customers. Start by clicking on Contact Us, then dig deeply to see if you can find a place where each company seeks or...
-
What does the Interpretation of Federal Financial Accounting Standards 9 discuss? When was the Interpretation promulgated? What was the purpose of the Interpretation?
-
What is the monetary unit assumption? What impact does inflation have on the monetary unit assumption?
-
Refer again to the gasoline sales time series data in Table. a. Use a weight of 1/2 for the most recent observation, 1/3 for the second most recent, and 1/6 for the third most recent to compute a...
-
7 uary 1 , 2 0 2 4 , Rick's Pawn Shop leased a truck from Corey Motors for a six - year period with an option to extend the for three years. Rick's had no significant economic incentive as of the...
-
The decomposition of nitrous oxide is found to proceed as follows: What is the order of this reaction with respect to N,O, and overall? +21/20 NO N + k[NO] -N0 = 1+ k[NO]
-
A 1100-K n-nonane thermally cracks (breaks down into smaller molecules) 20 times as rapidly as at 1000 K. Find the activation energy for this decomposition.
-
A freezer is maintained at 20F by removing heat from it at a rate of 75 Btu/min. The power input to the freezer is 0.70 hp, and the surrounding air is at 75F. Determine (a) The reversible power, (b)...
-
Explain how big data can be used in the field that you plan on working in when you graduate. 1. What type of data would you collect? 2. Would your data be structured or unstructured? 3. What would be...
-
Distinguish among the three principal types of auditors and indicate the types of audits each may perform.
-
An auditor who is testing IT controls in a payroll system would most likely use test data (discussed in Advanced Module 1 in Chapter 6) that contain conditions such as a. Deductions not authorized by...
-
A principal auditor decides not to refer to the audit of another CPA who audited a subsidiary of the principal auditor's client. After making inquiries about the other CPA's professional reputation...
-
Compute the missing amount in the accounting equation for each company (amounts in millions): Assets Liabilities TELUS.... ? $10,061 Scotiabank... 411,510 ? Shoppers Drug Mart. 5,644 2,434...
-
Use the same data as in the previous problem only suppose that the call price is $5 instead of $4.110. In previous problem Let S = $40, K = $40, r = 8% (continuously compounded), = 30%, = 0, T =...
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
a. Load the ICG on your computer and carry out the exercise. Performance number =________________________. (http://www.umich.edu/~elements/6e/icm/enzyme.html) b. Rederive Equation (9-9) assuming the...
-
Hydrogen radicals are important to sustaining combustion reactions. Consequently, if chemical compounds that can scavenge the hydrogen radicals are introduced, the flames can be extinguished. While...
-
The pyrolysis of acetaldehyde is believed to take place according to the following sequence: CH3CHOk1CH3+CHOCH3+CH3CHOk2CH3+CO+CH4CHO+CH3CHOk3CH3+2CO+H22CH3k4C2H6 a. Derive the rate expression for...
-
El Dorado Foods Inc. owns a chain of specialty stores in the Pacific Northwest. Recently, four of the stores have experienced declining profits due to market saturation in the area. As a result,...
-
PLEASE VERY URGENT manufacturing overhead is allocated at $ 7 per machine - hour based on fixed manufacturing costs of $ 5 2 5 , 0 0 0 7 5 , 0 0 0 machine - hours, which is the level Donaldson uses...
-
Zachary Company is a retail company that specializes in selling outdoor camping equipment. The company is considering opening a new store on October 1 , year 1 . The company president formed a...

Study smarter with the SolutionInn App


