For the reaction system of Example 9.4 (a) Find needed for 60% conversion of reactant using the
Question:
For the reaction system of Example 9.4
(a) Find needed for 60% conversion of reactant using the optimal temperature progression in the plug flow reactor.
(b) Also find the exit temperature of fluid from the reaction.
Example 9.4
Using the optimal temperature progression in a plug flow reactor for the reaction
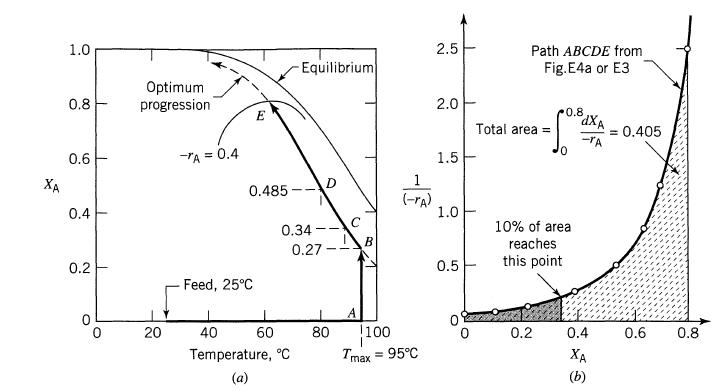
Transcribed Image Text:
XA 1.0 0.8 0.6 0.4 0.2 1 Optimum progression 20 -TA = 0.4 E 0.485 Feed, 25°C Equilibrium 0.34- 0.27 40 60 Temperature, °C 0 80 C A B (-rA) 1100 I Tmax = 95°C 2.5 2.0 1.5 1.0 0.5 0 Path ABCDE from Fig. E4a or E3 Total area = 0.8 0.2 0 10% of area reaches this point dXA -TA 0.4 XA = 0.405 0.6 0.8
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Discretize the optimal temperature progression This involves dividing the optimal temperature progression into a number of smaller segments each with ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Examples 9.4 through 9.7 illustrate the approach to problems dealing with non isothermal reactors. This approach to multistage operations of solid catalyzed reactions. To reinforce these concepts,...
-
Examples 9.4 through 9.7 illustrate the approach to problems dealing with non isothermal reactors. This approach to multistage operations of solid catalyzed reactions. To reinforce these concepts,...
-
For the optimum temperature progression in a plug flow reactor in Example 9.4 (C A0 = 4 mol/liter, F A0 = 1000 mol A/min, X A0 = 0.8, T = 5C, T max = 95C) and feed and product both at 25C, how much...
-
What are some ways that the antibody-protein complex can be visualized in ELISA or western blots?
-
On January 1, 2005, the controller of OBrian Manufacturing Company is planning capital expenditures for the years 20052008. The following interviews helped the controller collect the necessary...
-
Oak meanRapier means sword.s a tree that bears acorns. Determine whether the following are demonstrative definitions, enumerative definitions, definitions by subclass, synonymous definitions,...
-
Components of Internal Control. Internal control consists of five interrelated components. These are derived from the way management runs a business and are integrated with the management process....
-
The Green Thumb Gardener is a retail store that sells plants, soil, and decorative pots. On December 31, 2019, the firm's general ledger contained the accounts and balances that appear below....
-
\ table [ [ , A , B , c , D , E , F , G , H , , J , K , L , M , N , 0 , p ] , [ 1 , My Finances ] , [ 2 , , , , , , , , , , , , , , , , ] , [ 3 , ] , [ 4 , Income,$ 0 . 0 0 , $ 0 . 0 0 , $ 0 . 0 0 ,...
-
A pulse input to a vessel gives the results shown in Fig. P11.1. (a) Check the material balance with the tracer curve to see whether the results are consistent. (b) If the result is consistent,...
-
A liquid macrofluid reacts according to A R as it flows through a vessel. Find the conversion of A for the flow patterns
-
Each of 10 taste testers rated a new brand of barbecue sauce on a 10-point scale, where 1 = awful and 10 = excellent. Find the mode for the 10 ratings shown below. 8 7 9 6 8 10 9 9 5 7
-
When looking at your clients tax situation, you see they have paid a sizable amount of student loan interest of the following is not a good piece of advice to give them? O The tax law allows...
-
X Incorporated manufactures a single product called Product X and is sold at 100 per unit. Cost related to Product X at normal capacity of 625,000 direct labor hours are as follows: Direct Materials...
-
1- Describe the five-stage model of team development. 2- Describe the three dimensions of skill differentiation, authority differentiation, and temporal stability that comprise team structure....
-
George works as the office manager for Potato LLC, a medium-sized law firm. One afternoon, he went to the hardware store to purchase a few maintenance items for the office. While there, he also...
-
1.You are running a company with two divisions. One division is high risk, and the other division is low risk. Cost of Capital Expected Return on Proposed Projects. High risk division 10% 9% Low risk...
-
On March 20th of the current year, Stephen sold his common shares of Salt Ltd., an eligible small business corporation, for $800,000. His shares had an ACB of $100,000. On October 1st of the current...
-
1. Following are information about Alhadaf Co. Cost incurred Inventory Purchases Sales Adverting expense Salary Expense Depreciation Beginning Inventory Ending Inventory Amount 118,000 350.000 90,000...
-
You have two CSTRs and two PFRs, each with a volume of 1.6 m3. Use Figure 2-2(b) to calculate the conversion for each of the reactors in the following arrangements. 1. Two CSTRs in series. 2. Two...
-
The exothermic reaction of stillbene (A) to form the economically important trospophene (B) and methane(C), that is, A B+ C was carried out adiabatically and the following data recorded: The...
-
The financially important reaction to produce the valuable product B (not the real name) was carried out in Jesse Pinkmans garage (see Breaking Bad Season 3, Episode 7). This Breaking Bad,...
-
Yogi Company expects to sell 2 , 0 5 0 units of finished product in January and 2 , 4 0 0 units in February. The company has 2 7 0 units on hand on January 1 and desires to have an ending inventory...
-
Suppose that on March 1 , 2 0 2 5 , Amazon Company hires a new employee who will start to work on March 6 . The employee will be paid on the last day of each month. Should a journal entry be made on...
-
please fill this graph in and send it back to me thank you!

Study smarter with the SolutionInn App


