Substance A in the liquid phase produces R and S by the following reactions: The feed (C
Question:
Substance A in the liquid phase produces R and S by the following reactions: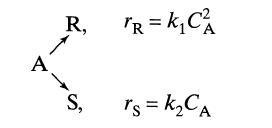
The feed (CAo = 1.0, CRo = 0, Cso = 0.3) enters two mixed flow reactors in series (τ1 = 2.5 min, τ2, = 10 min). Knowing the composition in the first reactor (CAI = 0.4, CRl = 0.2, CS1 = 0.7), find the composition leaving the second reactor.
Transcribed Image Text:
A R, S, TR = k₁C² rs = K₂ CA
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To find the composition leaving the second reactor we can use the given reactions and the concentrat...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Question 5 Draw a graph of a function f that satisfies the conditions below. On your sketch, indicate all relevant intercepts and equations of all asymptotes. (4) 1)f is even. 2) lim f(r) = 1 3) lim...
-
Here are five important questions for an exam in chemical engineering: Question 1: A liquid mixture containing 40% component A and 60% component B by mass is fed into a distillation column. The...
-
Dimethyl ether (DME) synthesis provides a simple prototype of many petrochemical processes. Ten tonnes (10,000 kg) per hour of methanol are fed at 25C. The entire process operates at roughly 10 bar....
-
Suppose that the owner of the restaurant in Problem 2.91 also wants to study the demand for dessert during the same time period. She decides that in addition to studying whether a dessert was...
-
For the coming year, India Ink Company anticipates a unit selling price of $400, a unit variable cost of $300, and fixed costs of $900,000. Instructions 1. Compute the anticipated break-even sales...
-
Professor Pearsons arguments in favor of the theory of evolution should be discounted. Pearson is a cocaine-snorting sex pervert and, according to some reports, a member of the Communist party....
-
Client Facilities Tour. When an auditor has accepted an engagement from a new client who is a manufacturer, it is customary for the auditor to tour the client's plant facilities. Discuss the ways in...
-
Transferred-in costs, FIFO method (continuation of 17-33). Refer to the information in Problem 17-33. Suppose that Larsen Company uses the FIFO method instead of the weighted-average method in all of...
-
Diddy Corp. stock has a beta of 1.3, the current risk-free rate is 7 percent, and the expected return on the market is 12.50 percent. What is Diddy's cost of equity? (Round your answer to 2 decimal...
-
Find C R , and C S , and for X A , = 0.9 in a mixed flow reactor. Liquid reactant A decomposes as follows: A feed of aqueous A (C A0 = 40 mol/m 3 ) enters a reactor, decomposes, and a mixture of A,...
-
Using separate feeds of A and B sketch the contacting pattern and reactor conditions which would best promote the formation of product R for the following systems of elementary reactions A + B R 2A ...
-
The carbon-14 decay rate of a sample obtained from a young tree is 0.260 disintegration per second per gram of the sample. Another wood sample prepared from an object recovered at an archaeological...
-
Social channels are no longer the biggest source of inspiration for customers to purchase a product. Discuss this statement in your own words.
-
How do power dynamics and social networks shape decision-making processes and resource allocation within complex organizational structures, such as matrix organizations or global conglomerates?
-
Our business: a social sharing platform Question: Be sure to consider and include all licensing requirements and any compliance requirements for your business and what your policies/procedures will...
-
What types of social media does Amazon participate in? Based on your observations, are there any potential security vulnerabilities that Amazon is exposed to based on the nature of their business or...
-
list a detailed benefits package for a general manager of a hotel and a house keeper
-
A Ltd. received the following dividend income during the current year: $1,000 of eligible dividends from a taxable Canadian corporation $1,000 of non-eligible dividends from a taxable Canadian...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
The reactions of ozone were studied in the presence of alkenes (from R. Atkinson et al., Int. J. Chem. Kinet., 15(8), 721). The data in Table P7-10C are for one of the alkenes studied, cis-2-butene....
-
Tests were run on a small experimental reactor used for decomposing nitrogen oxides in an automobile exhaust stream. In one series of tests, a nitrogen stream containing various concentrations of NO...
-
The thermal decomposition of isopropyl isocyanate was studied in a differential packed-bed reactor. From the data in Table P7-12A, determine the reactionrate-law parameters. TABLE P7-12A RAW DATA Run...
-
Olivia's dumplings has a current stock price of $ 5 6 , and pays a constant dividend of $ 3 . 2 5 per year, ever year, forever. What is the required rate of return for this stock?
-
Parker Plastic, Incorporated, manufactures plastic mats to use with rolling office chairs. Its standard cost information for last year follows: Standard Quantity Standard Price ( Rate ) Standard Unit...
-
Information for two alternative projects involving machinery investments follows. Project 1 requires an initial investment of $ 1 4 8 , 0 0 0 . Project 2 requires an initial investment of $ 1 3 3 , 0...

Study smarter with the SolutionInn App


