The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table
Question:
The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table P5.25 are obtained in steady-state runs. To obtain 75% conversion of reactant in a feed, CA0 = 0.8 mol/liter, what holding time is needed in a plug flow reactor?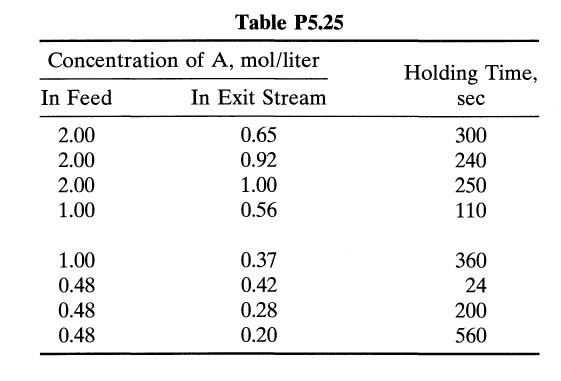
Transcribed Image Text:
Concentration In Feed 2.00 2.00 2.00 1.00 1.00 0.48 0.48 0.48 Table P5.25 of A, mol/liter In Exit Stream 0.65 0.92 1.00 0.56 0.37 0.42 0.28 0.20 Holding Time, sec 300 240 250 110 360 24 200 560
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To determine the required holding time in a plug flow reactor to achieve 75 conversion of reactant A ...View the full answer

Answered By

Prachi Goyal
No experience of tutoring and teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Repeat the previous problem but for a mixed flow reactor. Previous Problem The aqueous decomposition of A is studied in an experimental mixed flow reactor. The results in Table P5.25 are obtained in...
-
The aqueous decomposition of A produces R as follows: The following results are obtained in a series of steady state runs, all having no R in the feed stream. From this kinetic information, find the...
-
The kinetics of the aqueous-phase decomposition of A is investigated in two mixed flow reactors in series, the second having twice the volume of the first reactor. At steady state with a feed...
-
QUESTION 7 We discussed the selection sort sorting algorithm. What is the worst case time complexity of selection sort when sorting a list of n elements? o(lg n) O(n) O(n lg n) O(n) O(log n) O(1)
-
The manager for Tyler Bank and Trust has the following assets and liabilities to manage: If the manager wants a duration gap of 3.00, what level of saving accounts should the bank raise? Assume that...
-
In 2007 Hampton Office Supply began selling a new computer that carried a 2-year warranty against defects. Based on the manufacturer's recommendations, Hampton projects estimated warranty costs (as a...
-
Mercury is about \(3.114 \times 10^{7}\) miles from the sun. Neptune is about \(2.781 \times 10^{9}\) miles from the sun. How many times further is Neptune from the sun than Mercury?
-
The following information was abstracted from the accounts of the General Fund of the City of Rome after the books had been closed for the fiscal year ended June 30, 2012. During the year, purchase...
-
16. Examine the following t-bill quote. Your pointy haired boss has instructed you to purchase $1 million in these treasury bills. Days to Ask Maturity Date Maturity Bid Ask Yield Dec 31 '99 102 4.65...
-
The data in Table P5.28 have been obtained on the decomposition of gaseous reactant A in a constant volume batch reactor at 100C. The stoichiometry of the reaction is 2A R + S. What size plug flow...
-
A high molecular weight hydrocarbon gas A is fed continuously to a heated high temperature mixed flow reactor where it thermally cracks (homogeneous gas reaction) into lower molecular weight...
-
Determine the reactions for each structure. All dimensions are measured from the centerlines of members. 6 kips B 0.4 kip/ft 40' D E T 10' 20'
-
The number of combinations by: of taking objects out ofn objects is given In the Powerball lottery game the player chooses five numbers from 1 through 59, and then the Powerbail number from 1 through...
-
How do postmodernist perspectives challenge conventional notions of deviance by emphasizing the fluidity of meaning, the fragmentation of social norms, and the multiplicity of subcultural identities ?
-
Consider three vectors, A, B, and C such that A+B+C=-3.55 m. Find the magnitude of if A and B have components of: Ax -0.89 m Ay 0.67 m Bt 0.18 m By 0.92 m C= m Report your numerical answer below,...
-
Spelling Company has the following sales projection (in units) for the next six months: Feb: 7500 Mar: 8500 Apr: 6500 May: 10000 Jun: 8000 Jul: 5500 Each unit sells for $20. Spelling has prepared the...
-
5 3 To solve the rational equation how can the X-1 4 5 expression x-1 be rewritten using the least common denominator?
-
Explain the bundle-of-sticks idea as it relates to intellectual property.
-
Does log 81 (2401) = log 3 (7)? Verify the claim algebraically.
-
The irreversible gas-phase dimerization 2A A2 is carried out at 8.2 atm in a stirred contained-solids reactor to which only pure A is fed. There are 40 g of catalyst in each of the four spinning...
-
The second-order decomposition reaction A B + 2C is carried out in a tubular reactor packed with catalyst pellets 0.4 cm in diameter. The reaction is internal-diffusion-limited. Pure A enters the...
-
A first-order reaction is taking place inside a porous catalyst. Assume dilute concentrations and neglect any variations in the axial (x) direction. a. Derive an equation for both the internal and...
-
SYST 17796 ICE - SOLID/DRY + PATTERN This exercise is to be completed individually during class time. Students are allowed to discuss the exercise and provide assistance to one another, but each...
-
Lucy is the managing director of VitaJuice Ltd, a publicAustralian company which produces organic juices. The company has aconstitution, which states that the company may only purchaseoranges and l 2...
-
/** * @param handle - the User handle to check for * @return the first Reply object FROM a User with the passed handle. Return * null if no users match the passed handle....

Study smarter with the SolutionInn App


