The following data on an irreversible reaction are obtained with decaying catalyst in a batch reactor (batch-solids,
Question:
The following data on an irreversible reaction are obtained with decaying catalyst in a batch reactor (batch-solids, batch-fluid) What can you say about the kinetics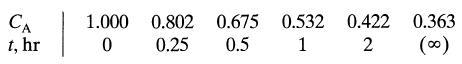
Transcribed Image Text:
CA t, hr 1 1.000 0 0.802 0.25 0.675 0.532 0.422 0.5 1 2 0.363 (00)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
The data you have provided shows the concentration of reactant A CA over time for an irreversible re...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The reversible catalytic reaction proceeds with decaying catalyst in a batch reactor (batch-solids, batchfluid). What can you say of the kinetics of reaction and deactivation from the following data:...
-
The kinetics of a particular catalytic reaction A R are studied at temperature T in a basket reactor (batch-solids and mixed flow of gas) in which the gas composition is kept unchanged, despite...
-
The kinetics of a particular catalytic reaction A R are studied at temperature T in a basket reactor (batch-solids and mixed flow of gas) in which the gas composition is kept unchanged, despite...
-
4. Papo and Pepe are two barbers from a small barbershop. Theyhave their two court chairs plus two waiting chairs. The followingresults were found: P0 = 1/16 P1 = 4/16 P2 = 6/16 P3 = 4/16 a. What is...
-
The following transactions were completed by Prairie Renaissance Inc., whose fiscal year is the calendar year: 2005 July 1. Issued $8,000,000 of 5-year, 8% callable bonds dated July 1, 2005, at an...
-
For 2020, the maximum percentage of Social Security benefits that could be included in a taxpayers gross income is: a. 0% b. 25% c. 50% d. 75% e. 85%
-
Capital plc carried on business in four product segments, namely aircraft design, hairdressing salons, import agencies and beauty products. The directors are now considering the dividend policy and...
-
On December 31, the payroll register of Hamstreet Associates indicated the following information: Wages and Salaries Expense $8,700.00 Employee Income Tax Payable 920.00 United Way Contributions...
-
Employers must identify the purposes for which they are collecting employee personal information without consent true or false?
-
A recycle reactor with very high recycle ratio is used to study the kinetics of a particular irreversible catalytic reaction, A R. For a constant flow rate of feed (' = 2 kg sec/liter) the...
-
In an automobile's catalytic converter, CO and hydrocarbons present in the exhaust gases are oxidized. Unfortunately the effectiveness of these units decreases with use. The phenomenon was studied by...
-
If 5 times a number is decreased by 4, the principal square root of this difference is 2 less than the number. Find the number(s).
-
For each of the users of accounting information, indicate whether they are an external decision maker (E) or an internal decision maker (I). a. Marketing manager b. Canada Revenue Agency c. Investor...
-
Using recent news headlines, describe an example of teaming in which a group of people came together briefly to solve a complex problem.
-
Solve the systems in Problems 13-16 by graphing, adding, substitution, Gauss-Jordan, or inverse matrix methods. \(\left\{\begin{array}{l}x+y+z=5 \\ x-2 y+z=-1 \\ 3 x+y-2 z=16\end{array} ight.\)
-
Explain the format and structure of the balance sheet of a typical limited company.
-
What is depreciation and what are the problems encountered in dealing with the depreciation of non-current assets?
-
A stock currently sells for $32.00. A 6-month call option with a strike of $35.00 has a premium of $2.27. Assuming a 4% continuously compounded risk-free rate and a 6% continuous dividend yield, what...
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
The elementary gas-phase reaction A + B 2C in Problem P11-8B is now continued and carried out in packed-bed reactor. The entering molar flow rates are F A0 = 5 mol/s, F B0 = 2F A0 , and F I = 2F A0...
-
Use the reaction data in Problems P11-4A and P12-7B for the case when heat is removed by a heat exchanger jacketing the reactor. The flow rate of coolant through the jacket is sufficiently high that...
-
Derive the energy balance for a packed-bed membrane reactor. Apply the balance to the reaction in Problem P11-5A A B + C for the case when it is reversible with KC = 1.0 mol/dm 3 at 300 K. Species C...
-
Calculate the expected costs when production is 5 , 4 0 0 units. ( Round per unit calculations to 2 decimal places, e . g . 1 5 . 2 6 and final answer to the nearest whole dollar, e . g . 5 , 2 7 5 ....
-
Lightfoot Inc., a software development firm, has stock outstanding as follows: 3 0 , 0 0 0 shares of cumulative preferred 4 % stock, $ 2 5 par, and 3 8 , 0 0 0 shares of $ 1 0 0 par common. During...
-
The following information relates to the debt imvestments of Ayayai Inc. during a recent year: On February 1 , the company purchased Gibbons Corp. 1 0 % bonds with a face value of $ 2 2 2 , 0 0 0 at...

Study smarter with the SolutionInn App


