We wish to treat 10 liters/min of liquid feed containing 1 mol A/liter to 99% conversion. The
Question:
We wish to treat 10 liters/min of liquid feed containing 1 mol A/liter to 99% conversion. The stoichiometry and kinetics of the reaction are given by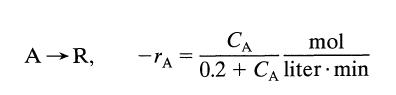
Suggest a good arrangement for doing this using two mixed flow reactors, and find the size of the two units needed. Sketch the final design chosen.
Transcribed Image Text:
A →R, -TA CA mol 0.2 + CA liter min
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Sure to tackle this problem we should first determine the type of reactor system arrangement that can achieve a 99 conversion of reactant A and then calculate the size volume of each reactor Based on ...View the full answer

Answered By

Evans Cherono
I am an Information Technology Graduate and willing to work on any computer science or IT work to ensure I do my best all the time.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
With the use of appropriate examples, contrast the use of milestones and critical paths for tracking of project progress. Furthermore, elaborate on the steps for the following: Viewing of critical...
-
The kinetics of the aqueous-phase decomposition of A is investigated in two mixed flow reactors in series, the second having twice the volume of the first reactor. At steady state with a feed...
-
Using a color indicator which shows when the concentration of A falls below 0.1 mol/liter, the following scheme is devised to explore the kinetics of the decomposition of A. A feed of 0.6 mol A/liter...
-
an 5 of 5 | Review Mode: Question 13 of 20 A survey asked 1,150 pe to choose their favorite laundry detergent from brands D, E, and F. Of the people surveyed, percent chose D as their favorite brand....
-
Distinguish between independent agents and exclusive agents.
-
Some nebulas are clouds of gas. Some clouds of gas are objects invisible to the naked eye. Therefore, some objects invisible to the naked eye are nebulas. Use the five rules to determine whether the...
-
Assume the same facts as in SE 6 except that the purchase of 100 percent of Winter Treats Corporation was for $120,000. After the purchase, what would be the amount, if any, of the following accounts...
-
1. Ultimately, who is responsible for decision making with regard to this situation? 2. What steps should be taken to remedy this situation? 3. Are there fair and ethical procedures to follow to...
-
Chip Company produces three products, Kin, Ike, and Bix. Each product uses the same direct material. Kin uses 3 . 4 pounds of the material, lke uses 3 . 1 pounds of the material, and Bix uses 5 . 5...
-
From steady-state kinetics runs in a mixed flow reactor, we obtain the following data on the reaction AR. Find the space time needed to treat a feed of C A0 = 100 mmollliter to 80% conversion (a) In...
-
At 100C pure gaseous A reacts away with stoichiometry 2A R + S in a constant volume batch reactor as follows: What size of plug flow reactor operating at 100C and 1 atm can treat 100 moles A/hr in a...
-
Use a graph or level curves or both to estimate the local maximum and minimum values and saddle point(s) of the function. Then use calculus to find these values precisely. f(x, y) = sin x + sin y +...
-
Review the discussion of ethical issues and the need theories of motivation. Discuss those issues with the goal of taking a strong position on them.
-
The accounting profession's commitment to achieving high quality in rendering professional services is demonstrated by the breadth and effectiveness of its multilevel regulatory framework. Required...
-
The criticisms raised about behavior modification. Discuss those criticisms. Do those criticisms limit the use of behavior modification in organizations?
-
Government regulation is one component of the profession's regulatory framework. Required a. Identify the regulatory organizations involved in government regulation and indicate the primary...
-
The AICPA's Division for CPA Firms and Quality Review Division play important roles in the profession's self-regulation activities. Required a. The activities of these two divisions are directed...
-
If XYZ does nothing to manage copper price risk, what is its profit 1 year from now, per pound of copper? If on the other hand XYZ sells forward its expected copper production, what is its estimated...
-
A copper rod of length L =18.0 in is to be twisted by torques T (see figure) until the angle of rotation between the ends of the rod is 3.08. (a) If the allowable shear strain in the copper is 0.0006...
-
Ethyl acetate is an extensively used solvent and can be formed by the vapor-phase esterification of acetic acid and ethanol. The chemical equation shows the formation of Ethyl acetate by the vapor...
-
The gas-phase reaction A + B C + D takes place isothermally at 300 K in a packed-bed reactor in which the feed is equal molar in A and B with CA0 = 0.1 mol/dm 3 . The reaction is second order in A...
-
Go to Professor Herzs Reactor Lab on the Web at www.reactorlab.net. Click on Download and then click on the English version link. Provide the information asked and then click Download. Select...
-
"This is really an odd situation," said Jim Carter, general manager of Highland Publishing Company. " We get most of the jobs we bid on that require a lot of press time in the Printing Department,...
-
Based on the information in the table, answer the following: Gross Revenue Total Operating Expense Net Operating Income Fixed Costs Largest Variable Cost ( amount ) :
-
The following data relate to labor cost for production of 2 0 , 0 0 0 cellular telephones: \ table [ [ Actual: , 8 , 4 5 0 h r s . at $ 2 2 . 5 0

Study smarter with the SolutionInn App


