An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. 2+
Question:
An electrochemical cell is based on these two half-reactions: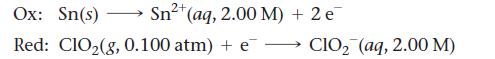
Calculate the cell potential at 25 °C.
Transcribed Image Text:
2+ Sn²+ (aq, 2.00 M) + 2 e Ox: Sn(s) Red: ClO₂(g, 0.100 atm) + e- CIO₂ (aq, 2.00 M)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Sure to calculate the cell potential Ecell for the electrochemical cell you can use the ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. Ox: Pb(s) Pb+ (aq, 0.10 M) + 2 e Red: MnO4 (aq, 1.50 M) + 4H* (aq, 2.0 M) + 3 e MnO (s) + 2 HO(1)
-
An electrochemical cell is constructed such that on one side a pure nickel electrode is in contact with a solution containing Ni2+ ions at a concentration of 3 10-3 M. The other cell half consists...
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
A UK company has a trading loss of 50,000 for the year to 31 March 2021. During the year, the company receives overseas property income (net of 40% withholding tax) of 12,000. Show the corporation...
-
1. A unit-level driver is consumed by a product each and every time that a. a batch of products is produced. b. a purchase order is issued. c. a unit is produced. d. a customer complains. e. none of...
-
Two coils are mutually coupled, with L1 = 25 mH, L2 = 60 mH, and k = 0.5. Calculate the maximum possible equivalent inductance if: (a) The two coils are connected in series (b) The coils are...
-
Financial statement disclosures include each of the following except: a. notes to the financial statements. b. parenthetical disclosures. c. supplementary information. d. promotional giveaways.
-
Santana Rey has consulted with her local banker and is considering financing an expansion of her business by obtaining a long-term bank loan. Selected account balances at March 31, 2012, for Business...
-
47 Required information [The following information applies to the questions displayed below.] Phoenix Company reports the following fixed budget. It is based on an expected production and sales...
-
What is a concentration electrochemical cell?
-
Explain the difference between a voltaic (or galvanic) electrochemical cell and an electrolytic cell.
-
Bob Jacobs opened an advertising agency. The following transactions occurred during January of the current year. Classify each transaction as an operating, an investing, or a financing activity. (a)...
-
During the 1990s the preppy Tommy Hilfiger brand unexpectedly became the uniform for teens in what became known as the urban wear market. Attempting to capitalize on this new found appeal among the...
-
Choose a brand or imagine what was your brand positioning? How is it differentiated from the competitors? Who was your specific target? Why did you select them?
-
"When you think of financial institutions, which ones come to mind?" What types of recall can this question be used to measure? Explain.
-
A new restaurant is planned that specializes in low-fat and low-carb healthy food. You have been asked to create a multiplatform campaign for the grand opening. Evaluate various media and platforms...
-
Due to the pandemic, people have been afraid to go to hospitals even for non-COVID-related illnesses. They think there's a high possibility of getting infected with COVID-19 when they are in hospital...
-
Sample: 20, 13, 4, 8,10 Find the population variance and standard deviation or the sample variance and standard deviation as indicated.
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
Describe what happens to the vapor pressure of water as the temperature increases.
-
Describe why it is important to consider NPSH when designing and operating a pumping system.
-
Find the available NPSH when a pump draws water at 140F from a tank whose level is 4.8 ft below the pump inlet. The suction line losses are 2.2 lb-ft/lb and the atmospheric pressure is 14.7 psia.
-
he Quick Corporation has 4,000 shares of 8%, 100 par value preferred stock and 100,000 shares of common stock outstanding. If net income at the end of the year is determined to be $375,000, what were...
-
Barnyard Traders uses a combination of shares and debt in their capital structure. The details are given below: There are 2 0 million R 1 0 . 8 0 ordinary shares in issue and the current market price...
-
Download the seasonally adjusted monthly labour force characteristics by province from StatisticsCanada for 25 to 54 years old people in last available month and the same month of last...

Study smarter with the SolutionInn App


