Consider the electrolytic cell: a. Label the anode and the cathode and indicate the halfreactions occurring at
Question:
Consider the electrolytic cell: 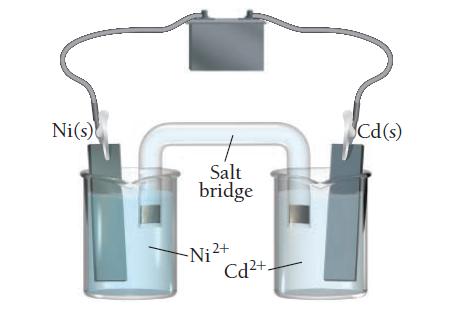
a. Label the anode and the cathode and indicate the halfreactions occurring at each.
b. Indicate the direction of electron flow.
c. Label the terminals on the battery as positive or negative and calculate the minimum voltage necessary to drive the reaction.
Transcribed Image Text:
Ni(s) Salt bridge Cd(s) Hi Ni ²+ Cd²+.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Anode Ni N...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ray Holt Corporation has retained you as a consultant on accounting policies and procedures. During 2019, the company engaged in a number of treasury stock transactions, having foreseen an...
-
26. Originally from England, Joy received her permanent Canadian resident status three years ago. She lives in Edmonton, where she works as a surgeon major hospital. She travels back to her home...
-
Consider the voltaic cell: a. Determine the direction of electron flow and label the anode and the cathode. b. Write a balanced equation for the overall reaction and calculate E cell . c. Label each...
-
Briefly explain the meaning of the following concepts and terms: greenhouse gases (GHGs), the IPCC, the little ice age, the Keeling graph, telekinetic property of the atmosphere, global weirding,...
-
Johanssen Company uses activity-based costing (ABC). Johansson manufactures outdoor water toys using two activities: plastic injection molding and decal application. Johanssons 2009 total budgeted...
-
The city mileage (in miles per gallon) for 24 family sedans Mileage (in miles per gallon) Frequency 22 27 . 16 28 33 . 2 34 39. 2 40 45 . 3 46 51 . 1 Approximate the mean of the frequency...
-
'The conditions under which budgetary control is most effective are precisely those under which it is of least value'. Specify the conditions that you think make for effective budgetary control, and...
-
Determine the error for each of the following forecasts. Compute MAD andMSE. Period Vale Forecast Error 202 191 173 169 202 192 181 174 172 174 179 189 198 211 4 175 182 196 204 219 227 10
-
5. After performing the translation described below, 6. After performing the translation described below, what will be the coordinate of point M? translation: (x, y)-(x-7, y + 1) what will be the...
-
Determine whether or not each metal, if coated onto iron, would prevent the corrosion of iron. a. Mg b. Cr c. Cu
-
Determine whether or not each metal, if coated onto iron, would prevent the corrosion of iron. a. Zn b. Sn c. Mn
-
Sam is having discussions with a Croatian company that might supply him with folding chairs and tables for sports events. Sam has been quoted a price of 100 kuna per chair and 400 kuna per table. As...
-
what does the number (1) (12) (11) (9) etc. means for a spreadsheet for the statement of cash flows? Balance 12/31/24 Satellite Corporation Worksheet for Preparation of Statement of Cash Flows for...
-
(a) Calculate the current ratio for each of the following competing companies. (Round your answers to 2 decimal places.) Current Assets Current Ratio Current Liabilities Edison $ 79,040 $ 32,000 MAXT...
-
Many countries argued that earnings management practices performed by a firm would increase the likelihood of fraud. 1- Discuss briefly the three earnings management methods (6 points) 2- Which...
-
The following data are available for Company A. Company A uses retail inventory method to determine ending inventory. Sales revenue is recorded net of employee discounts, which totaled $21,400 in the...
-
General Foundry, Incorporated, is a leading global manufacturer and marketer of branded consumer foods sold through retail stores. It recently disclosed the following information concerning the...
-
The consolidation that has developed at the retail end of many supply chains has been described as the Walmart effect. Why? What is the significance of retail consolidation for supply chains?
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
Obtain v 1 through v 3 in the circuit of Fig. 2.81. + 1 - ww "2 24 V V3 +) 10 V 12 V +
-
Describe a simple U-tube manometer.
-
Describe a differential U-tube manometer.
-
Comprehensive Problem (Algo) LO 11-1, 11-2, 11-3, 11-4 Benson Modems, Inc. acquired a subsidiary named Anywhere, Inc. (Al). Al manufactures a wireless modem that enables users to access the Internet...
-
1The building was purchased on May 3, 2009, and is depreciated to the nearest whole month using the straight-line method. Depreciation is based on a 15-year life, after which it will be demolished...
-
With respect to the adjustments made by the analyst in Country B related to property, plant, and equipment, answer the following: 1. Why does the adjustment for depreciation related to revalued...

Study smarter with the SolutionInn App


