Consider the molecular view of an electrochemical cell involving the overall reaction: Draw a similar sketch of
Question:
Consider the molecular view of an electrochemical cell involving the overall reaction:![]()
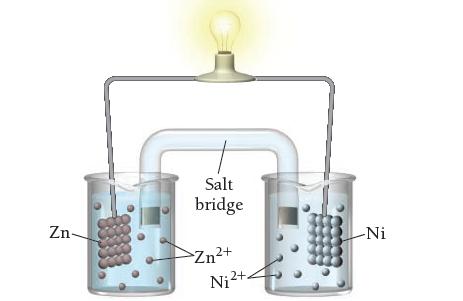
Draw a similar sketch of the cell after it has generated a substantial amount of electrical current.
Transcribed Image Text:
Zn(s) + Ni²+(aq) Zn²+(aq) + Ni(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Zns Ni2aq Zn2aq Nis This is a redox reactionwhere zinc metal is oxidized to zinc ionsand nickel ions ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For reaction, A + B 10L volumetric flask at 25C. If K =0.238 are available? C + D: Given that initial amounts of 0.68 mol of both A and B in a at equilibrium, how many moles of each reactant and...
-
The diagram shows an electrochemical cell involving two metal/metal-ion systems. The standard electrode potentials for the half-cells are: Ag + + e Ag E = +0.80 V Cu2 + + 2e Cu E = +0.34 V a....
-
a. Draw a diagram of an electrochemical cell consisting of a Mn 2+ /Mn half-cell and a Pb 2+ /Pb half-cell. b. Use the data in Appendix 2 to calculate the cell voltage. c. Which half-cell is the...
-
Economist Abba Lerner proposed a tariff on oil imports equal to 100 percent of the import price. This tariff is designed to reduce dependence on foreign sources as well as to discourage OPEC from...
-
Sorensen Manufacturing produces several types of bolts used in aircrafts. The bolts are produced in batches according to customer orders. Although there are a variety of bolts, they can be grouped...
-
Number of classes: 8 Data set: Number of hospitals in each state (a) An expanded frequency distribution, (b) A frequency histogram, (c) A frequency polygon, (d) A relative frequency histogram, and...
-
Explain the decision process you followed in making an important decision in the past which has now taken effect (e.g., the choice of a course of study and the university to attend; the choice of...
-
The Clarence Corporation has issued bonds that pay semiannually with the following characteristics: a. Calculate modified duration using the information provided. b. Explain why modified duration is...
-
You are holding a kite string in your hand. The angle of elevation from your hand to the kite is 53 and the distance to the kite is 300 feet. Your hand is 4 feet above the ground. How high is the...
-
Determine whether HI can dissolve each metal sample. If it can, write a balanced chemical reaction showing how the metal dissolves in HI and determine the minimum volume of 3.5 M HI required to...
-
Consider the molecular views of an Al strip and Cu 2+ solution. Draw a similar sketch showing what happens to the atoms and ions after the Al strip is submerged in the solution for a few minutes. Al...
-
How do the degree of confidence and the degree of precision differ?
-
Universal Travel uses the contribution margin income statement internally. Universal's first quarter results are as follows: Universal's relevant range is sales of between $130,000 and $650,000....
-
Rebecca holds 100 shares of Gotchas stock that she purchased for $3,400 several years ago. In a merger of Gotchas into Solis, Inc., Rebecca exchanges her 100 Gotchas shares for 1,000 Solis shares and...
-
According to Adam Smith, the free-market system will create maximum wealth for nations and benefit all members of society. According to Karl Marx, free market capitalism will enrich only the...
-
Tyler Company budgets the following unit sales for the next four months: April, 3,000 units; May, 4,000 units; June, 6,000 units; and July, 2,000 units. The company's policy is to maintain finished...
-
Jiminez, Incorporated, is an all-equity firm. The cost of the company's equity is currently 13.7 percent and the risk-free rate is 4.6 percent. The company is currently considering a project that...
-
A number of authors have observed that traditional, hierarchical organizations have changed in the current global economy. How have organizations changed? Why have they changed?
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
A buildup of fission products poisons a reactor, dropping the multiplication factor to 0.992. How long will it take the reactor power to decrease by half, given a generation time of 0.10 s?
-
The total thermal power generated in a nuclear power reactor is 1.5 GW. How much U-235 does it consume in a year?
-
New Hampshires Seabrook nuclear power plant produces electrical energy at the rate of 1.2 GW and consumes 1311 kg of U-235 each year. Assuming the plant operates continuously, find (a) its thermal...
-
Suppose a hardware engineering team runs a study looking for a statistically significant correlation between using circuit A in new computers and a system failure. Suppose that the team found a...
-
Windsor Ridge Corporation was organized on January 1, 2018. During its first year, the corporation issued 46,000 shares of $5 par value preferred stock and 480,000 shares of $1 par value common...
-
Assume that production and income are below their full employment levels. In the long run we would predict that aggregate demand will rise to restore full employment without any government...

Study smarter with the SolutionInn App


