Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following
Question:
Consider the reaction:
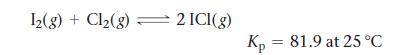 Calculate ΔGrxn for the reaction at 25 °C under each of the following conditions:
Calculate ΔGrxn for the reaction at 25 °C under each of the following conditions:
a. Standard conditions
b. At equilibrium
c. PICl = 2.55 atm; PI2 = 0.325 atm; PCl2 = 0.221 atm
Transcribed Image Text:
12(g) + Cl₂(g) = 2 ICI(g) Kp = 81.9 at 25°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The standard Gibbs free energy change AG ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P CH3OH = 1.0 atm; P CO = P H2 = 0.010 atm CO(g) +...
-
Ray Holt Corporation has retained you as a consultant on accounting policies and procedures. During 2019, the company engaged in a number of treasury stock transactions, having foreseen an...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
What problems may be encountered in making a comparative study of remuneration reports?
-
What is the difference between an activity flexible budget and a functional-based (traditional) flexible budget?
-
P(A) = 2/3, P(A') = 1/3, P(B | A) = 1/5, and P(B | A') = 1/2 Use Bayes Theorem to find P(A | B).
-
As a manager becomes more senior, it appears that he needs to rely more on formal information yet that his style of life encourages him to use it less. How would you explain this apparent paradox?
-
Nettles, King, and Tanaka are partners sharing income 3:2:1. After the firm's loss from liquidation is distributed, the capital account balances were: Nettles, $15,000 Dr.; King, $46,000 Cr.; and...
-
9 1 point LO What is the value of x? x 12 18 Type your answer... 1 point Find CD G 2x-7 E 11
-
Explain the difference between macrostates (external arrangements of particles) and microstates (internal arrangements of particles).
-
Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73. Problem 73 Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction....
-
Using the same bond as in Question 9, assume that you believe interest rates are going to increase. What do you think will happen to the value of the bond? What if the bond was a putable convertible...
-
Fuzzy Button Clothing Company's weighted average cost of capital is 10%, and project Beta has the same risk as the firm's average project. Based on the cash flows, what is project Beta's NPV?
-
A few weeks after meeting with your firm, James Clements received a text from Brad Raymore apologizing for his absence. With the construction industry booming, Brad had been working a lot of...
-
On August 1, 2021, Lotore, Inc., sold equipment and accepted a six-month, 9%, $10,000 note receivable. Lotore's year-end is December 31. How much interest revenue should Lotore accrue on December 31,...
-
Suppose the real rate is 1.9 percent and the inflation rate is 3.1 percent. What rate would you expect to see on a Treasury bill? Real rate Inflation rate 1.90% 3.10% Complete the following analysis....
-
How to write statement of our intention to progress into masters of professional accounting advance?
-
1. What are the benefits of a sales career? 2. How can GPI Procurement Services sales representatives add value for customers?
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
Name a type of compressor often used for pneumatic fluid power systems.
-
The intake duct to a fan consists of intake louvers, 5.8 m of square duct (800 800 mm), a sudden contraction to a 400-mm-diameter round duct, and 9.25 m of the round duct. Estimate the pressure at...
-
The electric field of an electromagnetic plane wave is given in SI units by (a) What is the wave?s angular frequency? (b) Write an expression for vector k. (c) What is the value of k? (d) Determine...
-
abc equipment purchased equipment on 10/1/2023 that costs 12000 by issuing note payable. note is a three years 8% note and equipment will last in 5 years with no salvage value. calculate the ajusting...
-
Generally, the receipt of corporate bond interest is more valuable than preferred dividends to corporate investors. True or False
-
ABC Corporations Certificate of Incorporation was revoked by SEC for failure to comply with reportorial requirements. It has no creditor and its lone asset is a parcel of land. Out of 9 directors,...
DiFiores Atlas Of Histology With Functional Correlations 12th Edition - ISBN: 1451113412 - Free Book

Study smarter with the SolutionInn App


