Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom. H
Question:
Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom.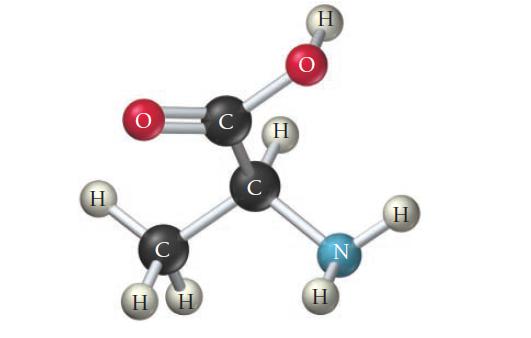
Transcribed Image Text:
H Η Η Η H H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
H H H ...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose you are the head of the marketing department for an on-demand video streaming service. A registered user pays a monthly subscription fee to access the platforms entire library. Your research...
-
Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom. H 0 0 Z H H H H
-
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping...
-
. 4. Bank overdrafts repayable on * 1 point demand may be included in the cash and cash equivalent balance. True O False
-
Dilts Corporation sells three different models of mosquito zapper. Model A12 sells for $50 and has variable costs of $40. Model B22 sells for $100 and has variable costs of $70. Model C124 sells for...
-
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction. Discuss.
-
Information technology is continually changing the nature of accounting and the role of accountants. Write a two-page report describing what you think the nature of the accounting function in a large...
-
Hayes Industries purchased the following assets and constructed a building as well. All this was done during the current year. Assets 1 and 2: These assets were purchased as a lump sum for $100,000...
-
Trixie has nicely asked her children to put their toys away. Her children do not do so, therefore Trixie decides that she will store all of their toys in the basement for a month. Trixie is using a....
-
Sketch the bonding molecular orbital that results from the linear combination of two 1s orbitals. Indicate the region where interference occurs and state the kind of interference (constructive or...
-
Write a hybridization and bonding scheme for each molecule or ion. Sketch the structure, including overlapping orbitals, and label all bonds using the notation shown in Examples 11.6 and 11.7. a. SO...
-
Use the method of Examples 7 and 8 to find the Jordan normal form J of each coefficient matrix A given in Problems 23 through 32 (respectively). Example 7 In Example 3 we saw that the matrix ^=[03] A...
-
How do preventive controls differ from detective controls? Are they mutually exclusive, redundant, or complementary? Provide three examples to support your response.
-
List the methods of internal controls by category.
-
A small private company, Agchem Ltd, is involved in the research and development of chemicals used in the agricultural industry. The company is based in Drumnadrochit, a small remote Scottish...
-
List at least 10 guidelines for achieving effective, crisp, clear writing.
-
What is meant by management override? Should it be prohibited? How should its occurrence be monitored?
-
Bevil Industries is deciding whether to automate one phase of its production process. The manufacturing equipment has a six-year life and will cost $900,000. Projected net cash inflows are as...
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Referring back to the multiple antenna system on p. 456, compute the angular separation between successive lobes or principal maxima and the width of the central maximum.
-
Examine the setup of Fig. 10.3 in order to determine what is happening in the image space of the lenses; in other words, locate the exit pupil and relate it to the diffraction process. Show that the...
-
Consider the case of single-slit Fraunhofer diffraction. Calculate the ratio of the irradiance of the central maximum to the irradiance of the first secondary maximum on either side of it. Check your...
-
Julia and Hannah are 50% partners in North Star General Partnership. Each has 100,000 basis in her partnership interest. During the current year, Julia receives 20,000 of cash distributions and...
-
5) (3 pts) You buy a $1000 face value bond that matures in 3 years, pay a 5% annual coupon and are priced at $970. a. What is the Yield to maturity of these bonds? YTM = % b. What is your realized...
-
1 ) For each fund ( ABNDX , MSFT , MA , NVDA ) provide or attach documentation ( hand or send it to me ) that shows: - the assets under management ( AUM ) - the size and style category and / or...

Study smarter with the SolutionInn App


