Consider the titration curves (labeled a and b) for two weak bases, both titrated with 0.100 M
Question:
Consider the titration curves (labeled a and b) for two weak bases, both titrated with 0.100 M HCl.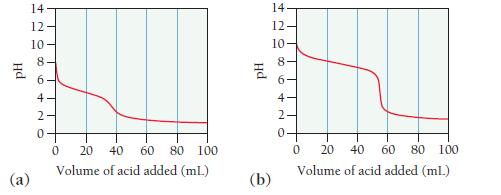
i. Which base solution is more concentrated?
ii. Which base has the larger Kb?
Transcribed Image Text:
Hd (a) 14 == 208 12 10 6 4 N 0 0 20 40 60 80 100 Volume of acid added (mL) PH (b) 14. 12- 10- 00 10 8 6- 4- 2. 0 0 20 40 60 80 100 Volume of acid added (ml.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The base solution in curve a is more concentrated than the base solution in curv...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A mountain-climbing expedition establishes two intermediate camps, labeled A and B in the drawing, above the base camp. What is the magnitude (r of the displacement between camp A and camp B? 4900 m...
-
A chemical company produces amongst its product range two industrial cleaning fluids, A and B. These products are manufactured jointly. Total sales are expected to be restricted because home trade...
-
The figure in the preceding exercise shows the pH curves for the titrations of six different acids with NaOH. Make a similar plot for the titration of three different bases with 0.10 M HCl. Assume...
-
How does ESMA support national regulators in enforcing the application of IFRS?
-
What is cycle time? Velocity?
-
In an RLC circuit, R = 20 , L = 4 H and C = 1 F. The circuit is magnitude-scaled by 10 and frequency-scaled by 10 5. Calculate the new values of the elements.
-
Kate Miller owns a dance studio in Los Angeles, California. Students can buy access to the dance classes by paying a monthly fee. Unfortunately, many of Kates students are struggling actors and...
-
Sunny Valley Resort has owned 80 percent of Mountain Lodging, Inc. since Mountain Lodging's inception. The condensed consolidated balance sheets of Sunny Valley Resort at December 31,2012 and 2011...
-
Patrick Mahomes, a corporation, created a sports equipment company on July 1, 2025. Patrick Mahomes Sports Equipment is a wholesaler that specializes in sports equipment sold to colleges,...
-
Phenolphthalein has a pK a of 9.7. It is colorless in its acid form and pink in its basic form. For each of the values of pH, calculate [In ]/[HIn] and predict the color of a phenolphthalein...
-
Calculate G and K for each reaction the group created in Question 143. For one of the reactions, explain how the sign or magnitude of each quantity (Ecell, G, and K) is consistent with the fact that...
-
Evaluate a failed organizational change, identify a theory that could have been used to develop the change, and apply that theory to the failed change. The paper must follow these standards: Your...
-
There are four points A (2, 6), B(6, 4), C(4, 0) and D(0, 2) on the coordinate plane. Identify the pairs of parallel and perpendicular lines
-
Summer Day Inc. manufactures two models of its hammock, the Superior and the Deluxe. The Superior model requires 15, 0 0 0 direct labour hours and the Deluxe requires 45, 0 0 0 direct labour hours....
-
Solve the compound inequality for x. 2x 24 or 3x-1>8
-
The following table provides a probability distribution for the random variable x. x369 f(x) .25 .50 .25 a. Compute E(x), the expected value of x. b. Compute 2, the variance of x (to 1 decimal). c....
-
Sally has highlighted concerns that Lakeside is susceptible to a risk of sending tow vehicles to jobs that do not exist. This is because the business has yet to develop a reliable way of validating...
-
We have all heard of the Old Faithful geyser in Yellowstone National Park. However, there is another, less famous, Old Faithful geyser in Calistoga, California. The following data represent the...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Figure P.11.49 shows a transparent ring on an otherwise opaque mask. Make a rough sketch of its autocorrelation function, taking l to be the center-to-center separation against which you plot that...
-
Return to Eq. (12.21) and separate it into two terms representing a coherent and an incoherent contribution, the first arising from the superposition of two coherent waves with irradiances of |? ? 12...
-
Referring to the previous two problems with the cosine grating oriented horizontally, make a sketch of the electric-field amplitude along y' with no filtering. Plot the corresponding image irradiance...
-
how is a budgeted functional income statement for future years important
-
Research has observed that the more fire trucks that show up to put out a fire, the more damage there is to the building that was on fire. There is a _______________ correlation between the number of...
-
What are the portfolio weights for a portfolio that has 132 shares of Stock A that sell for $42 per share and 112 shares of Stock B that sell for $32 per share? (Do not round intermediate...

Study smarter with the SolutionInn App


