Consider the zinc blende structure in Figure 13.16. What type of structure would result if the remaining
Question:
Consider the zinc blende structure in Figure 13.16. What type of structure would result if the remaining tetrahedral sites in the unit cell were also filled with cations?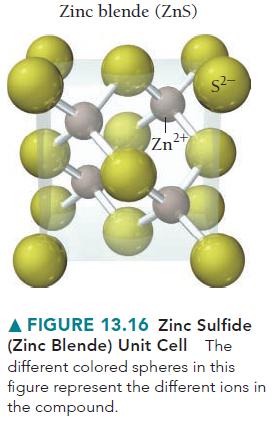
Transcribed Image Text:
Zinc blende (ZnS) Zn²+ S²- A FIGURE 13.16 Zinc Sulfide (Zinc Blende) Unit Cell The different colored spheres in this figure represent the different ions in the compound.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
If the remaining tetrahedral sites in the zinc blende structure were also filled with cations the re...View the full answer

Answered By

Swati gupta
Professionally I am going to become a Cost and Management Accountant very soon. I am a fresher in the area of tutoring but I am always admired and praised by my friends for teaching them. They always ask me to start giving tutoring services professionally. With the help of this platform, I am looking forward to give my best.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
what does the following print #include 2 class Parent{ public: Parent () { cout
-
Show that the unit cells for CaF 2 and TiO 2 in Figure 12-50 are consistent with their formulas. Figure 12-50 = Zn+ (a) Unit cell of ZnS, the zinc blende structure = $- = Ti4+ = Ca+ = 0- (c) Unit...
-
Cadmium telluride, CdTe, takes the zinc blende structure (Figure 12.26) with a unit cell edge length of 6.49 . There are four cadmium atoms and four tellurium atoms per unit cell. How many of each...
-
Happy Hands Company has net profit margin 5.51%, total assets turnover 0.63, and equity multiplier 2.77. What is its return on shareholder equity (ROE) using DuPont analysis method
-
Max E. Pass, Jr., and his wife, Martha N. Pass, departed in an aircraft owned and operated by Mr. Pass from Plant City, Florida, bound for Clarksville, Tennessee. Somewhere over Alabama the couple...
-
The Tech Mech Company produces and sells 6,000 modular computer desks per year at a selling price of $500 each. Its current production equipment, purchased for $1,500,000 and with a five-year useful...
-
Low gearing means: a. A company depends largely on long-term loans b. A company has few, if any, long-term loans c. A company cannot pay a dividend d. A company has high interest payments
-
ANALYSIS OF PROFITABILITY Based on the financial statement data in Exercise 24-1B, compute the following profitability measures for 20-2 (round all calculations to two decimal places): (a) Profit...
-
93 liters is the same as how many gallons? Hint: 1 L 0.26 gal Round your answer to the nearest tenth.
-
The shaded area in the following graph represents the feasible region of a linear programming problem whose objective function is to be maximized. Label each of the following statements as True or...
-
Classify each of the following as a component of a silicate ceramic, an oxide ceramic, or a nonoxide ceramic. a. B 4 C b. Mg 2 SiO 4 c. MoSi 2
-
Consider the rock salt structure in Figure 13.15. What type of structure would result if all the anions were somehow removed, leaving only cations? Sodium chloride (NaCl) CI Na+ A FIGURE 13.15 Sodium...
-
When the Fed increased the monetary base between 2008 and 2014, which component of the monetary base increased most: banks reserves or currency? What happened to the reserves that banks borrowed from...
-
Please use current tax rates, exemptions, and laws. Be sure to show ALL of your work so that I can give you partial credit. Use 2 0 2 3 tax law. Use these instructions: 2 0 2 3 Instruction 1 0 4 0 (...
-
It is easy for anybody to delegate tasks in an office, but effective leadership is more than that. Prominent leaders have significant impacts on the team members they manage and their company....
-
Find a song whose lyrics he/she feel convey something about one's(personality, perception of self, belief systems, ethnicity, nationality etc).ok, so you find asong. 2.Post a link to the video &...
-
Write a short(5-7 sentences long) narrative about an experience you have had with climate change. Your story can be about an extreme weather event that you experienced, a time when the power went...
-
Anita is working for a garment factory stitching material on an hourly basis.She is paid based on a piecework compensation plan whereby the base rate is RM5 per hour. Within each hour, Anita must...
-
Find the critical value (or values) for the t test for each. a. n = 10, = 0.05, right-tailed b. n = 18, = 0.10, two-tailed c. n = 6, = 0.01, left-tailed d. n = 9, = 0.025, right-tailed e. n = 15,...
-
If the cylinder described in Problem 21.3 were initially heated to 500F, how long would it take for the center of the cylinder to cool to 240F if it were constructed of a. Copper? b. Brass? c. Nickel?
-
At what temperature is the rms of Ar equal to that of SF 6 at 298 K? Perform the same calculation for mp .
-
Determine the temperature at which ave for Kr is equal to that of Ne at 298 K.
-
The probability that a particle will have a velocity in the x direction in the range of v x0 and v x0 is given by m? /2kT dv% 1/2 f(-v, s V, s V, ) = 2rkT 1/2 v mv /2kT dv x rkT
-
Analice cual de estas empresas est orientada ms orientada a un futuro en ciudades y comunidades sostenibles? Grupo de opciones de respuesta La composicin de los productos de BIOFASE es de 60%...
-
In airline financial terms, the abbreviation "FPA" can refer to which of the following? Group of answer choices (A) Fixed Position Aloft (B) Financial Planning and Analysis (C) Forecast of Penalty...
-
To calculate the amount of the sale (before sales tax) divide the total sales price by 1.08. Merchandise listed on the sales invoice: Stock Number Item Quantity Net Cost Per Unit AU150 JET SKI ULTRA...

Study smarter with the SolutionInn App


