Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from
Question:
Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from an orbital in n = 6 to an orbital in n = 5.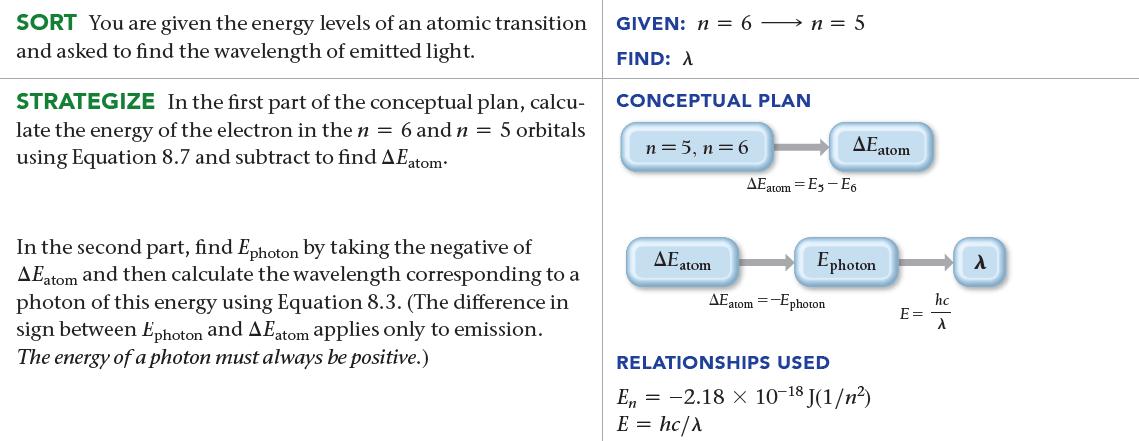
Transcribed Image Text:
SORT You are given the energy levels of an atomic transition and asked to find the wavelength of emitted light. STRATEGIZE In the first part of the conceptual plan, calcu- late the energy of the electron in the n = 6 and n = 5 orbitals using Equation 8.7 and subtract to find AEatom In the second part, find Ephoton by taking the negative of AEatom and then calculate the wavelength corresponding to a photon of this energy using Equation 8.3. (The difference in sign between Ephoton and AEatom applies only to emission. The energy of a photon must always be positive.) GIVEN: n = 6→ n = 5 FIND: A CONCEPTUAL PLAN n = 5, n = 6 AE atom AEatom AE atom-Es-E6 Ephoton AEatom =-Ephoton RELATIONSHIPS USED En = -2.18 × 10-18 J(1/n²) E = hc/λ hc E = - λ λ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
AEatom Es E6 181218 1018 1018 2 218 x 1018 1 5 6 26644 x ...View the full answer

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from an orbital in n = 5 to n = 4.
-
Calculate the wavelength of the light emitted when an electron in a hydrogen atom makes each transition and indicate the region of the electromagnetic spectrum (infrared, visible, ultraviolet, etc.)...
-
An electron in a hydrogen atom is excited from the ground state to the n = 4 state. Comment on the correctness of the following statements (true or false). (a) n = 4 is the first excited state. (b)...
-
According to a study conducted by the Gallup organization, the proportion of Americans who are afraid to y is 0.10. A random sample of 1100 Americans results in 121 indicating that they are afraid to...
-
Elbert Company was organized on January 1. During the first year of operations, the following plant asset expenditures and receipts were recorded in random order. Debits 1. Cost of real estate...
-
Polar Company purchased a building with an expected useful life of 40 years for $600,000 on January 1, 2016. The building is expected to have a residual value of $40,000. Required: a. Give the...
-
Using the cost and revenue information shown for DeKalb, Inc., on the next page, determine how the costs, revenue, and cash flow items would be affected by three possible exchange rate scenarios for...
-
Cedar Grove Industries produces and sells a cell phone-operated home security control. Information regarding the costs and sales of security controls during May 2017 are provided below. Unit selling...
-
Click the icon to view the data and two-way analysis of variance results. rst test for an interaction between the two factors. Determine the null and alternative hypotheses. Choose the correct answer...
-
You are a profitable conglomerate thinking about getting into the gelati business by acquiring the firm Alati Gelati (AG). Current info for you, AG and their similar comp is listed below. You...
-
What determines the color of a colored object? Explain why grass appears green.
-
What values of l are possible for n = 3? (a) 0 (or s) (b) 0 and 1 (or s and p) (c) 0, 1, and 2 (or s, p, and d) (d) 0, 1, 2, and 3 (or s, p, d, and f)
-
Electric car maker Tesla is recruiting Nevadans with a high school diploma or equivalent to work in the battery gigafactory the company is building. Explain how Teslas huge project will influence...
-
How do implicit cognitive biases and stereotyping manifest in organizational decision-making processes, and what mechanisms can be employed to mitigate their impact ?
-
Question 1 Sagbeug Consider the following information. Each day a butcher buys a 200 kg pig for R3 000. The pig can be processed to yield the following three products: Pork chops Bacon Ham Selling...
-
The number of combinations by: of taking objects out ofn objects is given In the Powerball lottery game the player chooses five numbers from 1 through 59, and then the Powerbail number from 1 through...
-
How do postmodernist perspectives challenge conventional notions of deviance by emphasizing the fluidity of meaning, the fragmentation of social norms, and the multiplicity of subcultural identities ?
-
Consider three vectors, A, B, and C such that A+B+C=-3.55 m. Find the magnitude of if A and B have components of: Ax -0.89 m Ay 0.67 m Bt 0.18 m By 0.92 m C= m Report your numerical answer below,...
-
In examining a simple random sample of 100 sales invoices from several thousand such invoices for the previous year, a researcher finds that 65 of the invoices involved customers who bought less than...
-
If M = 7, s = 2, and X = 9.5, what is z?
-
Why can you conclude that the energy of the anti-bonding MO in H + 2 is raised more than the energy of the bonding MO is lowered?
-
The total energy of a molecule is lowered if the orbital energy of the anti-bonding MO is negative, and raised if the orbital energy of the anti-bonding MO is positive. The zero of energy is the...
-
Consider the molecular electrostatic potential map for the LiH molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Manufacturing Company Balance Sheet Partial balance sheet data for Upper Crust Company at August 31 are as follows: Finished goods inventory $16,500 Prepaid insurance 16,800 Accounts receivable...
-
Lou Barlow, a divisional manager for Sage Company, has an opportunity to manufacture and sell one of two new products for a five- year period. His annual pay raises are determined by his division's...
-
Current Attempt in Progress Cullumber Bicycle Company manufactures its own seats for its bicycles. The company is currently operating at 100% capacity. Variable manufacturing overhead is charged to...

Study smarter with the SolutionInn App


