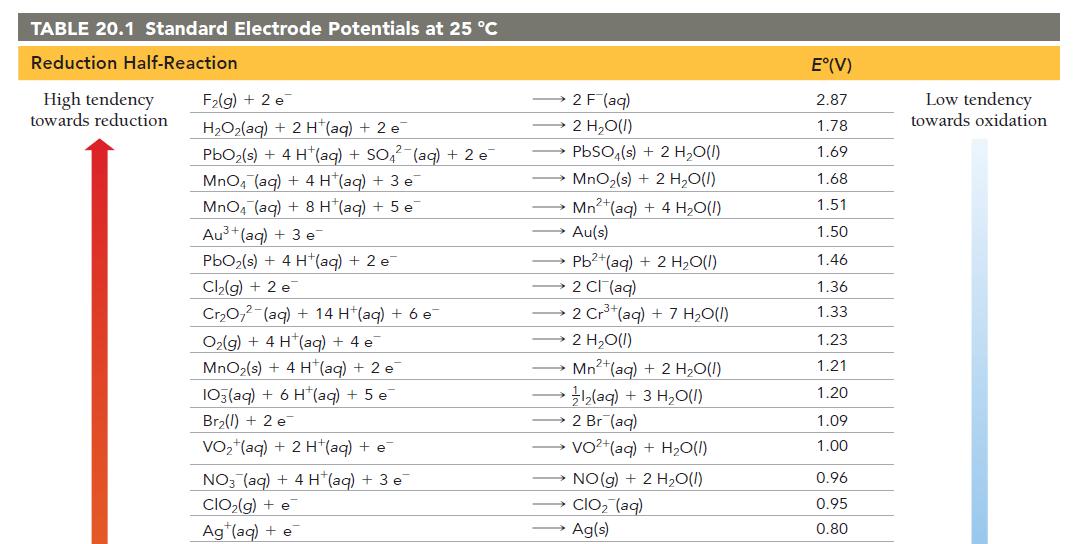
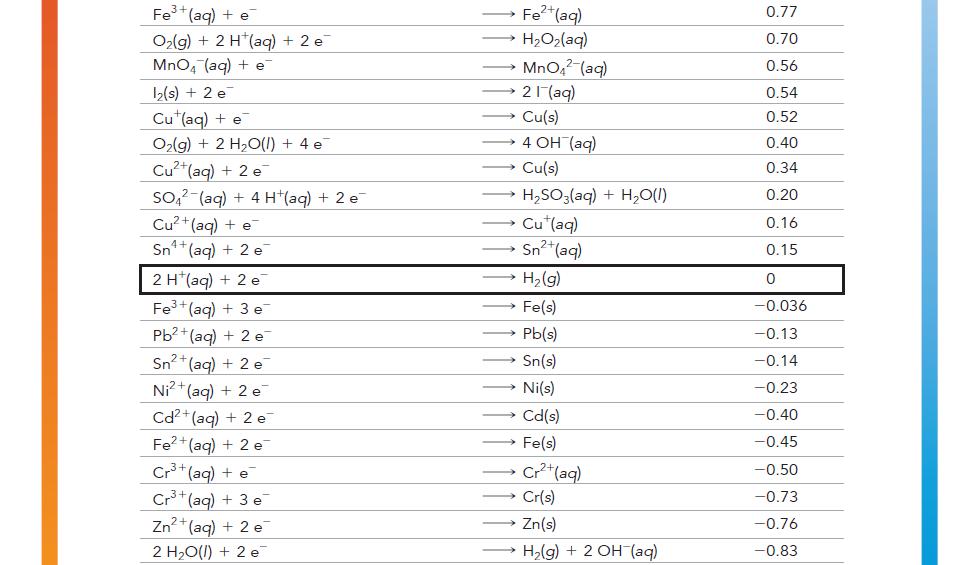
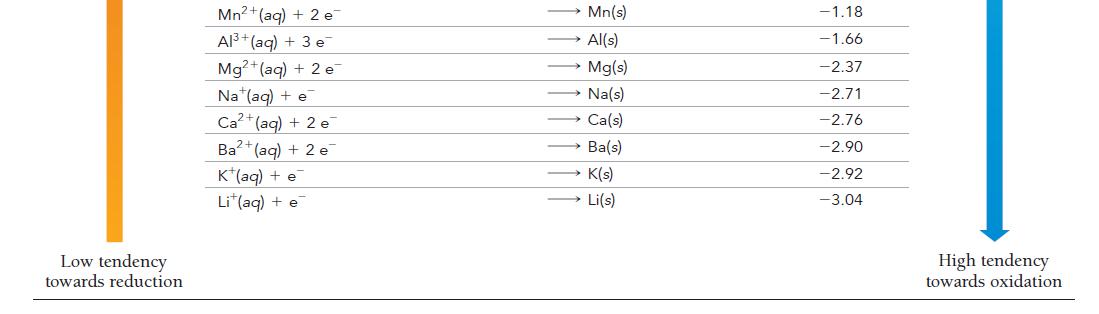
Have each group member select a half-reaction from Table 20.1. Each member should calculate the standard cell
Question:
Have each group member select a half-reaction from Table 20.1.
Each member should calculate the standard cell potential of an electrochemical cell formed between each member’s halfreaction and the half-reaction of each of the other group members.
For each pair of half-reactions, write the overall balanced chemical reaction that will be spontaneous.
Transcribed Image Text:
TABLE 20.1 Standard Electrode Potentials at 25 °C Reduction Half-Reaction High tendency towards reduction F₂(g) + 2 e H₂O₂(aq) + 2 H(aq) + 2 e PbO₂ (s) + 4 H¹(aq) + SO₂² (aq) + 2 e MnO4 (aq) + 4 H (aq) + 3 e MnO4 (aq) +8 H*(aq) + 5 e Au³+ (aq) + 3 e PbO₂ (s) + 4 H(aq) + 2 e Cl₂(g) + 2 e Cr₂O2 (aq) + 14 H+(aq) + 6 e O₂(g) + 4 H(aq) + 4 e MnO₂(s) + 4 H+ (aq) + 2 e 103(aq) + 6 H(aq) + 5 e Br₂() +2 e VO₂ (aq) + 2 H+(aq) + e NO3(aq) + 4 H (aq) + 3 e ClO₂(g) + e Ag (aq) + e 2 F (aq) 2 H₂O(l) PbSO₁(s) + 2 H₂O(1) MnO₂ (s) + 2 H₂O(1) Mn2+ (aq) + 4H₂O(l) Au(s) Pb2+(aq) + 2 H₂O(l) 2 Cl(aq) 2 Cr³+(aq) + 7 H₂0(1) 2 H₂O(1) Mn+ (aq) + 2 H₂O(1) }(aq) + 3 H,O(I) 2 Br (aq) → VO+(aq) + H,O() NO(g) + 2 H₂O(1) CIO₂ (aq) Ag(s) E°(V) 2.87 1.78 1.69 1.68 1.51 1.50 1.46 1.36 1.33 1.23 1.21 1.20 1.09 1.00 0.96 0.95 0.80 Low tendency towards oxidation
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Have each group member select a halfreaction from Table 201 E...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate G and K for each reaction the group created in Question 143. For one of the reactions, explain how the sign or magnitude of each quantity (Ecell, G, and K) is consistent with the fact that...
-
Calculate the standard cell potential of the lead storage cell whose overall reaction is PbO2(s) + 2HSO4 (aq) + 2H+(aq) + Pb(s) 2PbSO4(s) + 2H2O(l)
-
stock options are no longer as valuable and employees in startups are losing out to founders and early investors for all of these reasons except: there is higher employee turnover, less of a delay in...
-
By applying modern technology to agriculture, the United States has become the most productive food-producing nation in the world. The secret to solving the world food security problem lies in...
-
Minihan Company had the data below for its most recent year, ending December 31: Required: Prepare a performance report that shows the variances on an item-by-itembasis. Actual costs: Indirect labor...
-
The company in Example 2 identifies 240 adults who are heavy smokers. The subjects are randomly assigned to be in a treatment group or in a control group. Each subject is also given a DVD featuring...
-
Grupo Bimbo, Krispy Kreme, and Tastykake are all competitors in the global marketplace. Key comparative figures (in millions) for these companies' recent annual accounting periods follow. Required 1....
-
Describe the key activities performed by IS professionals in each step of the SDLC.
-
The diagram below shows an angle with an unknown measure of & radians and & circle with & Faulds 2.3 CH Jong The terminal point is 1.18 cm above the circle's center. 23 cm 18 cm 10 a. What is the...
-
The cell Pt(s) |Cu + (1 M), Cu 2+ (1 M) II Cu+(1 M) |Cu(s) has E = 0.364 V. The cell Cu(s) |Cu 2+ (1 M) II Cu+(1 M) |Cu(s) has E = 0.182 V. Write the cell reaction for each cell and explain the...
-
An electrochemical cell has a positive standard cell potential but a negative cell potential. Which statement is true for the cell? a. K> 1;Q> K c. K> 1; Q < K b. K < 1;Q> K d. K < 1; Q < K
-
Determine the smallest lever force P needed to prevent the wheel from rotating if it is subjected to a torque M. The coefficient of static friction between the belt and the wheel is s. The wheel is...
-
Julian Thomas, who is single, goes to graduate school part-time and works as a waiter at the Bay Grill in San Francisco. During 2018, his gross income was $20,700 in wages and tips. He has decided to...
-
January Taxes February Taxes March Taxes Q 1 Earnings FWT $ 1 2 , 4 0 0 $ 1 2 , 7 2 0 $ 1 2 , 1 2 0 $ 3 3 1 , 2 0 0 Social Security $ 7 , 5 7 9 . 5 0 $ 7 , 2 4 0 . 3 6 $ 7 , 9 9 0 . 1 3 $ 3 6 7 , 9 0...
-
nollonut Use the graphs below for questions 3 and 4. Graph A 8x Graph B srov twoted adi havia noinnut fra s most annitumo lin 3. If the function in Graph A is horizontally stretched by a factor of 3,...
-
APC Wood is a business that sells wood to both domestic and overseas customers. All the income and expenses include GST if necessary. Below are the business details and income and expenses report for...
-
2/3 of A is equal to B. 30% of B is equal to C. The ratio of C:D is 3:5. If B is equal to 16, find the difference between A and D.
-
Why is there so much interest in radio-frequency identification? What supply chain benefits does RFID facilitate?
-
What are the key dimensions of critical thinking 2. Watch the NBC Learn video on Diet Scams. What types of claims are made in this video Are they valid Elaborate on your responses. Discuss this video...
-
Use the 2.16g/cm 3 density of NaCl to calculate the ionic spacing r 0 in the NaCl crystal.
-
Express the 7.84-eV ionic cohesive energy of NaCl in kilocalories per mole of ions.
-
Lithium fluoride, LiF, has the same crystal structure as NaCl and therefore has essentially the same Madelung constant a. Its ionic cohesive energy is -10.5 eV and the value of n in Equation 37.4 is...
-
Ignoring taxes, which of the following situations will cause comprehensive income to decrease? A. The amortization of an actuarial pension loss. B. An unrealized gain on an available-for-sale...
-
Job costing uses subsidiary ledgers to track the costs of individual jobs. True False
-
n 2023, Joshi has $2,500 short-term capital loss, $6,500 0%/15%/20% long-term capital gain and $3,500 qualified dividend income. Joshi is single and has other taxable income of $7,500. Which of the...

Study smarter with the SolutionInn App


