The solubility of phenol in water at 25 C is 87 g/L. The solubility of naphthol at
Question:
The solubility of phenol in water at 25 °C is 87 g/L. The solubility of naphthol at the same temperature is only 0.74 g/L. Examine the structures of phenol and naphthol shown here and explain why phenol is so much more soluble than naphthol.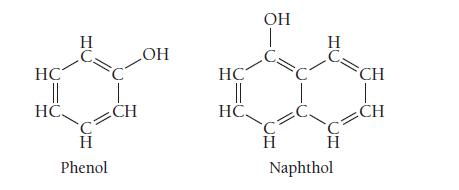
Transcribed Image Text:
HC || HC Н C. Н Phenol I CH OH HC || HC. OH н H Н Naphthol CH CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Phenol is more soluble in water than naphthol because it is more polar The hydroxyl group OH in phen...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following table presents the solubilities of several gases in water at 25 C under a total pressure of gas and water vapor of 1 atm. (a) What volume of CH 4 (g) under standard conditions of...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Why is it that I got 0 Row with nothing showing up? Is there a flow in my logic? Using AdventureWorks DW2017, list all the Canadian customers who have spent more than $5,000 total. Show Customerkey,...
-
Identify several types of printers. What are the advantages and disadvantages of each type?
-
What are the long-term consequences for Doles decision not to focus primarily on taste?
-
Because of a salary cap, National Basketball Association teams are not allowed to exceed a certain annual limit in total player salaries. Suppose the Toronto Raptors had scheduled salaries exactly...
-
A nursing home employs attendants who are needed around the clock. Each attendant is paid the same, regardless of when his or her shift begins. Each shift is 8 consecutive hours. Shifts begin at 6...
-
3) Listed below are the ages of randomly selected race car drivers. Construct a 99% confidence interval estimate of the mean age of all race car drivers. 30, 31, 33, 34, 45, 29, 38, 39, 33, 21, 27,...
-
Potassium perchlorate (KClO 4 ) has a lattice energy of -599 kJ/mol and a heat of hydration of -548 kJ/mol. Find the heat of solution for potassium perchlorate and determine the temperature change...
-
An aqueous CaCl 2 solution has a vapor pressure of 81.6 mmHg at 50 C. The vapor pressure of pure water at this temperature is 92.6 mmHg. What is the concentration of CaCl 2 in mass percent?
-
Why can a relatively small number of stock appreciation rights prove to be a material drain on future earnings and cash of a company?
-
The developer of this course had a very small wager placed on the University of Kansas to win the NCAA Division 1 Basketball National Championship. The ticket (contract) had been purchased in July...
-
Safeflower Systems allocates manufacturing overhead based on machine hours. Each connector should require 10 machine hours. According to the static budget, Safeflower Systems expected to incur the...
-
Using the CAPM formula listed below: Required rate of return = ERI=Rf+i(ERM-Rf) where:ERi-expected return of investment Rf=risk-free rate Bi-beta of the investment (ERm-Rf)=market risk premium...
-
3. Compute the worst case time complexity of the following algorithm. for i = 1 to n do for j = 21-1 to 2 - 1 do print (i, j).
-
Differentiate. y=25x-1 dy dx || log (5x+1)
-
You are shown a coin that its owner says is fair in the sense that it will produce the same number of heads and tails when flipped a very large number of times. a. Describe an experiment to test this...
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
Making use of the expression I(y) = I 0 e -y [4.78] for an absorbing medium, we define a quantity called the unit transmittance T 1 . At normal incidence, Eq. (4.55), T = I t /I i , and thus when y =...
-
Show that at normal incidence on the boundary between two dielectrics, as n ti S 1, R 0, and T 1. Moreover, prove that as n ti 1, R 0, and T 1 Moreover, prove that as n ti 1, R || 0, R 0, T...
-
Derive the expressions for r ¥ and r || given by Eqs. (4.70) and (4.71). cos 6; (n sin 0;)'/ cos 0; + (n sin 0;)'/2 (4.70) ni cos 0; (ni sin e,)'/2 n cos e; + (ni sin0;}'/2 and (4.71)
-
To seek innocent spouse relief, separation of liability relief, or equitable relief, which form should a taxpayer submit to the IRS? Form 843. Form 2210. Form 8379. Form 8857
-
SANDHILL MANAGEMENT CONSULTANTS Consolidated Income and Retained Earnings Statementfor the Year Ended December 31, 2024 Revenues 76,700 Operating Expenses including Depreciation of 2,100 francs...
-
You look at another report and find wages for the bar for another period were $11,421. At that time, you recall you operated staff in the bar on a 9% of sales basis. What sales did you need to make,...

Study smarter with the SolutionInn App


