Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write
Question:
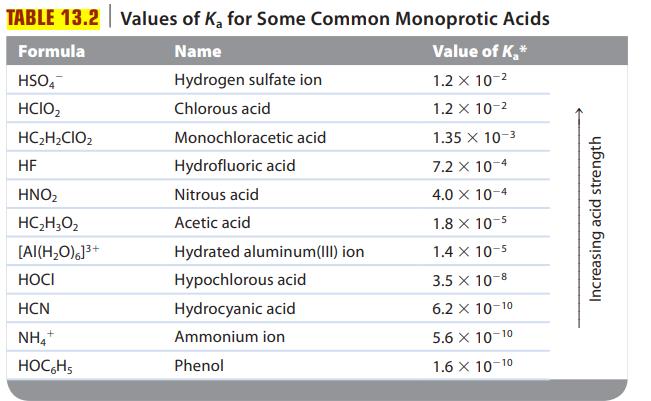
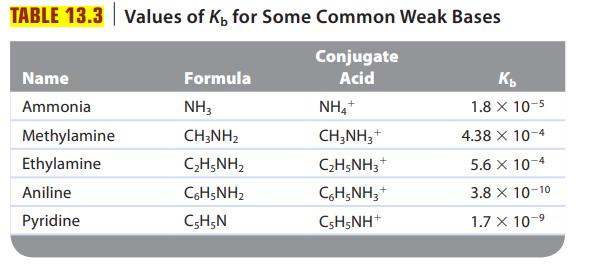
Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka and Kb values are found in Tables 13.2 and 13.3.
Transcribed Image Text:
TABLE 13.2 Values of K₂ for Some Common Monoprotic Acids Name Value of K₂* Hydrogen sulfate ion 1.2 x 10-² Chlorous acid 1.2 x 10-² 1.35 x 10-3 7.2 x 10-4 4.0 X 10-4 Formula HSO4 HCIO₂ HC₂H₂CIO₂ HF HNO₂ HC,H,Oz [AI(H₂O)]³+ HOCI HCN NH4+ HOCHS Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol 1.8 x 10-5 1.4 x 10-5 3.5 x 10-8 6.2 X 10-10 5.6 X 10-10 1.6 X 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Solutions of the following salts are acidic basic or neutral Salt Solution Equation NaHS Basic NaHS H2O HS Na H3O NH4Cl Acidic NH4Cl NH4 Cl NaCH3COO B...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Lets take a look at the extensive career of Helen Frankenthaler (1928-2011), who found a way to incorporate all of the innovative techniques artists were experimenting with starting in 1945. Her...
-
Will 0.10 M solutions of the following salts be acidic, basic, or neutral? a. Ammonium bicarbonate b. Sodium dihydrogen phosphate c. Sodium hydrogen phosphate d. Ammonium dihydrogen phosphate e....
-
Decide whether solutions of the following salts are acidic, neutral, or basic. a. Ammonium acetate b. Anilinium acetate
-
Which of the following would be the most frequently occurring daily transaction in a retail shop? (a) Paying salary to the sales assistant (b) Sale of goods (c) Payment of rent for the shop premises...
-
James Lillards first wife had a child whom James adopted when he married that childs mother. James fathered other children with her until they divorced in the early 1970s. In 1975, James married his...
-
Refer to the information in Exercise and assume instead that double-declining depreciation is applied. Compute the machine's payback period (ignore taxes). (Round the payback period to three...
-
Customized Stairs Co. of Poway designs and builds factory-made premium wooden stairs for homes. The manufactured stair components (spindles, risers, hangers, hand rails) permit installation of stairs...
-
Central Incorporated has two items in inventory as of December 31, 2011. Each item was purchased for $40. Company management chose to write down Item # 1 $28, which at year end was assessed to be its...
-
Factory Overhead Cost Variance Report Tannin Products Inc. prepared the following factory overhead cost budget for the Trim Department for July of the current year, during which it expected to use...
-
Which series has the highest beta. BraveNewCoin Liquid Index for Bitcoin 1D BNC Trading Brave Ne Yellow Green Blue Orange
-
Consider a solution of an unknown salt having the general formula BHCl, where B is one of the weak bases in Table 13.3. A 0.10-M solution of the unknown salt has a pH of 5.82. What is the actual...
-
The K b values for ammonia and methylamine are 1.8 10 -5 and 4.4 10 -4 , respectively. Which is the stronger acid, NH 4 + or CH 3 NH 3 + ?
-
Show that even if TS > 0, FS may or may not be greater than 0.
-
Amtrak's 20th-Century Limited is on route from Chicago to New York at 120km/h when the engineer spots a cow on the track. The train brakes to a halt in 1.3min, stopping just in front of the cow. a)...
-
2.(7.5 pts) Find the center of mass of the two-dimensional plate bounded by the curves: y=4x2 and y = 0 with density y at (x, y).
-
A frustrated dad is dragging their 36 kg child who is sitting on top of a 4.5 kg sled up a snow covered 30 degree incline. If the coefficient of friction between the steel runners and the snow is...
-
Explains and assesses suitable approaches to business process management analysis and design for the British Government.
-
An experimental rocket plane lands and skids on a dry lake bed. If it's traveling at 75.0 m/s when it touches down, how far does it slide before coming to rest? Assume the coefficient of kinetic...
-
The Internet Use data file on the text CD contains data on the number of individuals in a country with broadband access and the population size for each of 33 nations. When using population size as...
-
Starr Co. had sales revenue of $540,000 in 2014. Other items recorded during the year were: Cost of goods sold ..................................................... $330,000 Salaries and wages...
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed by an epoxide, followed by heating in aqueous acid: Using this process, identify what...
-
Predict the major product of the following transformation. CO2ET C10H100 Heat
-
How does the answer came from? Consider the following data for the assembly division of Gannett Watches Company:The assembly division uses the weighted-average method of process costing.Compute the...
-
Accounting I1) FEC LLC. made sales of $10,000 plus sales tax at 20% to a customer. CUSTOMERJONES paid 80% of the total liability in cash and agreed to pay the remaining after 15days. Choose the...
-
El objetivo de esta actividad es que los estudiantes investiguen y comprendan el anlisis de depreciacin en el contexto de la ingeniera econmica, as como su relacin con los impuestos. Debern presentar...

Study smarter with the SolutionInn App


