Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a.
Question:
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution.
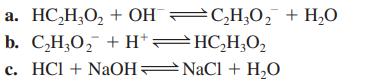
Transcribed Image Text:
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Image a chemical equation for a chemical reaction HC2H2O2 OH C2H2O2 H2O The equil...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Calculate the value for the equilibrium constant for each of the following aqueous reactions. a. NH 3 + H 3 O + NH 4 + H 2 O b. NO 2 - + H 3 O + HNO 2 + H2O c. NH 4 + CH 3 CO 2 - NH3 + CH 3 CO 2 H...
-
a. Calculate the value of Ka for the following acids: i. 0.0200 mol dm 3 2-aminobenzoic acid, which has a pH of 4.30 ii. 0.0500 mol dm 3 propanoic acid, which has a pH of 3.10 iii. 0.100 mol dm 3...
-
A firm is engaged in the production of two types of products. The first product (A) earns a profit of $4 per unit and the second product (B) earns $7 per unit. The sales force requires that at least...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Richard Vanderbrooks home in New Orleans, Louisiana, was insured through Unitrin Preferred Insurance Co. His policy excluded coverage for, among other things, flood, surface water, waves, tidal...
-
SBD Phone Company sells its waterproof phone case for $90 per unit. Fixed costs total $162,000, and variable costs are $36 per unit. Determine the (1) Contribution margin per unit (2) Break-even...
-
Jung Company's break-even sales are \($600,000\). Assuming fixed costs are \($240,000\), what sales volume is needed to achieve a target net income of $56,000?
-
Gatlin Company issued $300,000, 8%, 15-year bonds on December 31, 2011, for $288,000. Interest is payable annually on December 31. Gatlin uses the straight-line method to amortize bond premium or...
-
Q. Which of the following statements is FALSE regarding cost behaviors (within relevant range)? Multiple Choice Total variable cost increases and decreases in proportion to changes in activity level....
-
(a) A horizontal steel I-beam of cross-sectional area 0.041m2 is rigidly connected to two vertical steel girders. If the beam was installed when the temperature was 30oC, what stress is developed in...
-
Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH). b. Rank the four titrations in order of...
-
Consider a solution that contains both C 5 H 5 N and C 5 H 5 NHNO 3 . Calculate the ratio [C 5 H 5 N]/[C 5 H 5 NH + ] if the solution has the following pH values: a. pH = 4.50 b. pH = 5.00 c. pH =...
-
Discuss the assumptions and advantages of multistage compression.
-
Should Peoples Federal Savings have hedged its September 1 savings certificate rollover? What would you have advised Mr. Myers to do on August 6? How should Mr. Myers explain his future losses to the...
-
In the executive management model, what are the two possible types of committee a CISO should chair?
-
Determine the power series 4x representation of f(x) and 1 + x2 find the radius of convergence and interval of convergence for this power series. Radius of convergence [Select] Interval of...
-
What are the challenges in implementing comprehensive DEI programs, and how do these programs affect organizational performance and innovation ?
-
4. Two skaters are at a circular outdoor skating rink with a radius of 25 m. As they cross the centre of the rink, one skater grabs onto the other one. Prior to the collision one skater (with a mass...
-
The figure shows recent data on x = the number of televisions per 100 people and y = the birth rate (number of births per 1000 people) for six African and Asian nations. The regression line,...
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
The acetoacetic ester synthesis cannot be used to make 3,3-dimethyl-2-hexanone. Explain why not.
-
The product of a Dieckmann cyclization can undergo alkylation, hydrolysis, and decarboxylation. This sequence represents an efficient method for preparing 2-substituted cyclopentanones and...
-
Identify the major product formed when each of the following compounds is treated with Et2CuLi followed by mild acid. (a) (b) (c) CN
-
Gencon Contracting has a blanket contractors equipment policy. If Gencon acquires some new equipment, what kind of coverage is probably provided on this equipment? (Search Chapter 9) a. No coverage...
-
rewrite "I believe that my high standard of ethical conduct is shown in my assignment that I have submitted. I give credit where credit is due, and if I have questions concerning APA guidelines, I...
-
In December 2022, Surf Ltd received a claim from one of its customers commencing a lawsuit alleging faults in the construction of a bridge in a housing estate. This lawsuit amounts to a total of $2.5...

Study smarter with the SolutionInn App


