Given the following equilibrium constants at 427C, Determine the values for the equilibrium constants for the following
Question:
Given the following equilibrium constants at 427°C,
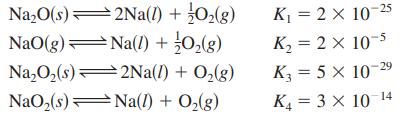
Determine the values for the equilibrium constants for the following reactions:
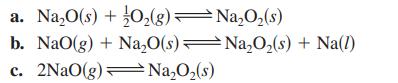
Transcribed Image Text:
2Na(1) + O₂(g) Na₂O(s) NaO(g) Na(1) + O₂(g) Na₂O₂ (s) 2Na(1) + O₂(g) NaO₂(s)Na(l) + O₂(g) K₁ = 2 × 10-25 K₂= 2 × 10-5 K3 = 5 x 10-2 29 K₁ = 3 × 10-14 -
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To determine the equilibrium constants for the reactions youre ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Given the following equilibrium constants at 427oC, Na2O(s) 2Na(l) + 12 O2(g) K1 = 2 10-25 NaO(g) Na(l) + l2 O2(g) K2 = 2 10-5 Na2O2(s) 2Na(l) + O2(g) K3 = 5 10-29 NaO2(s) Na(l) + O2(g) K4 = 3...
-
The following equilibrium constants were determined at 1123 K: Write the equilibrium constant expression KP, and calculate the equilibrium constant at 1123 K for C(s) + CO2(g)--2CO(g) CO(g) + Cl2(g)...
-
The following equilibrium constants have been determined for hydrosulfuric acid at 25°C: Calculate the equilibrium constant for the following reaction at the same temperature: H2S(aq) H(aq)HS...
-
What line of code can you add to disable all logging messages in your program?
-
Lazio never reported the income or paid the sales taxes from the Sylvan Beach pizzeria and never obtained workers compensation insurance for its employees. How might these facts have affected the...
-
The city of Gruberville is considering whether to build a new public swimming pool. This pool would have a capacity of 800 swimmers per day, and the proposed admission fee is $6 per swimmer per day....
-
Study Exhibit 10-2. Suppose the annual cash inflow will be 2 , 500 r a t h e r t h a n 2 , 500 r a t h e r t h a n 2 , 500 r a t h e r t h a n 2 , 500 r a t h e r t h a n 2,000. What is the internal...
-
Hewlett-Packard (HP) is considered one of the best-managed and most innovative companies in the world. It continually has shown an ability to adapt to global competitive challenges through technical...
-
For fiscal year 2020, Page Products had income as follows: Sales $55,400,000 Less: Cost of goods sold 38,900,000 Selling and administrative expense 5,770,000 Interest expense 1,040,000 Income before...
-
Yin & Yang Video has the following unadjusted trial balance as of January 31, 2010: The debit and credit totals are not equal as a result of the following errors: a. The balance of cash was...
-
For the reaction: K = 1.8 10 -7 at a certain temperature. If at equilibrium [O 2 ] = 0.062 M, calculate the equilibrium O 3 concentration. 30(g) 203(g)
-
At a particular temperature, K = 1.00 10 2 for the reaction In an experiment, 1.00 mole of H 2 , 1.00 mole of I 2 , and 1.00 mole of HI are introduced into a 1.00-L container. Calculate the...
-
The veterinarian instructs you to call Mrs. Smith and have her change Sweeties dose from (1) 100 mg capsule PO SID to (1) 100 mg capsule PO TID 3 d. What will you tell Mrs. Smith?
-
Question 15 of 25. In order to comply with federal banking regulations, you must complete and pass this training course prior to undertaking any bank representative related activities. OTRUE FALSE...
-
Suppose that a group is assigned a group presentation for a class. Group members briefly discuss to divide up the content, and then each individual researches and writes his or her portion of the...
-
Find amount of (A) net price and (B) trade discount dollar amount. (2 marks) Stove: List $1,400 Chain discount 13/10/4 A____________ B. ____________
-
A goldfish is swimming inside a spherical bowl of water, which has an index of refraction of n = 1.333. Suppose the goldfish is at p = 10.0 cm from the wall of the bowl, which has a radius |R| = 15.0...
-
How was the PV formula on excel used to calculate these? 22-Jan Cash $ Discount on Bond Payable SASA 68,917.00 $ 6,083.00 Bond Payable $ 75,000.00
-
Use the points from the previous exercise with x = 3, 5, 6, 7. a. Find the z -scores on x and on y for each point. Comment on how zx and zy relate to each other for each point. b. Compute r using the...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
Pyridine undergoes electrophilic aromatic substitution at the C3 position. Justify this regiochemical outcome by drawing resonance structures of the intermediate produced from attack at C2, at C3,...
-
Predict the product obtained when pyrrole is treated with a mixture of nitric acid and sulfuric acid at 0C.
-
Draw the structure of each of the following compounds: (a) Cyclohexylmethylamine (b) Tricyclobutylamine (c) 2,4-Diethylaniline (d) (1R,2S)-2-Methylcyclohexanamine (e) ortho-Aminobenzaldehyde
-
hello , can you please help me answer those a question correctly, thank you. Saved Help Save & Exit Subn Keisha owns 100% of DEF Company and has a basis ofof $12,000. Keisha has a: $10,000 in its...
-
Accountants that are focused on issuing financial statements to serve the dicision making needs of external users of the business are called
-
Under the default provisions, the financial rights between member-management owners and manager-management owners is: Question 2 options: a) immaterial. b) material, giving a higher percentage of...

Study smarter with the SolutionInn App


