How is acid strength related to the value of Ka? What is the difference between strong acids
Question:
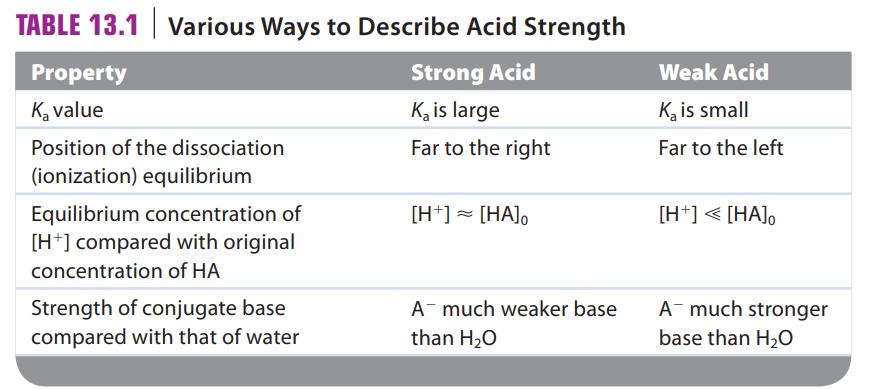
How is acid strength related to the value of Ka? What is the difference between strong acids and weak acids (see Table 13.1)? As the strength of an acid increases, what happens to the strength of the conjugate base? How is base strength related to the value of Kb? As the strength of a base increases, what happens to the strength of the conjugate acid?
Transcribed Image Text:
TABLE 13.1 Various Ways to Describe Acid Strength Strong Acid K₂ is large Far to the right Property K₂ value Position of the dissociation (ionization) equilibrium Equilibrium concentration of [H+] compared with original concentration of HA Strength of conjugate base compared with that of water [H+] = [HA]。 A much weaker base than H₂O Weak Acid K₂ is small Far to the left [H+] < [HA], A much stronger base than H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
a The acid dissociation constant Ka is a measure of how strongly an acid dissociates in waterThe hig...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
Draw a class diagram that reflect the following Java code segment as shown in Figure 4. public abstract class Vehicle { private int numberofWheels; public Vehicle () { this (0); } protected Vehicle...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Match each of the key terms with the definition that best fits it. ________ The process of ensuring that only authorized changes are made to a system. Here are the key terms from the chapter. The...
-
What would the order of inheritance have been if Ramish had died intestate? In June 2007, Bernard Ramish set up a $48,000 trust fund through West Plains Credit Union to provide tuition for his...
-
Warrenia has two regions. In Oliviland, the marginal benefit associated with pollution cleanup is MB = 300 10Q, while in Linn eland, the marginal benefit associated with pollution cleanup is MB =...
-
Castro Cabinets Company designs and builds upscale kitchen cabinets for luxury homes. Many of the kitchen cabinet and counter arrangements are custom made, but occasionally the company does mass...
-
An article published in Accounting Horizons describes various techniques that companies use to manage their earnings. Required: In your library or from some other source, locate the article How Are...
-
1 TB MC Qu. 07-111 (Algo) Jasper makes... Jasper makes a $42.000, 90-day, 9% cash loan to Clayborn Company Jasper's entry to record the collection of the note and interest at maturity should be (Use...
-
Professional and Scientific Staff Management (PSSM) is a unique type of temporary staffing agency. Many organizations today hire highly skilled technical employees on a short-term, temporary basis to...
-
Define or illustrate the meaning of the following terms: a. Amphoteric b. K w reaction c. K w equilibrium constant d. pH e. pOH f. pK w Give the conditions for a neutral aqueous solution at 25C, in...
-
At 35C, K = 1.6 10 -5 for the reaction If 2.0 moles of NO and 1.0 mole of Cl 2 are placed into a 1.0-L flask, calculate the equilibrium concentrations of all species. 2NOCI(g)2NO(g) + Cl(g)
-
Using probabilistic arguments, prove the identity \((A>a)\) : \[ 1+\frac{A-a}{A-1}+\frac{(A-a)(A-a-1)}{(A-1)(A-2)}+\ldots+\frac{(A-a) \ldots 2 \cdot 1}{(A-1) \ldots(a+1) a}=\frac{A}{a} \] An urn has...
-
Discuss the effectiveness of pharmacological interventions in the management of extreme stress and their potential side effects?
-
You throw a baseball at a 45 angle to the horizontal, aiming at a friend who's sitting in a tree a distance above level ground. At the instant you throw your ball, your friend drops another ball. The...
-
Duo8, Inc. is a U.S. corporation that was formed in MA in 1942. Amanda, one of the internal accountants at Duo8, Inc., has recently discovered that they have an individual investor, Clarke Johnson,...
-
You decide to finance a $ 1 3 , 0 0 0 car at 3 % compounded monthly for four years. Which steps would you take to find the total interest paid over the life of the loan?
-
2. A thin, semi-infinite wire with a constant linear charge density A extends from -00 < x < 0. A point charge q is placed at a position x = a with a > 0. Find an expression for the magnitude of the...
-
Describe a situation in which it is inappropriate to use the correlation to measure the association between two quantitative variables.
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
Draw the major product that is expected when each of the following compounds is treated with excess methyl iodide followed by aqueous silver oxide and heat: (a) Cyclohexylamine (b)...
-
Propose a synthesis for the following transformation (be sure to count the carbon atoms): Br
-
Compound A is an amine that does not possess a chirality center. Compound A was treated with excess methyl iodide and then heated in the presence of aqueous silver oxide to produce an alkene. The...
-
The European Commission rebuked Greece in 2010 for falsifying public finance data. Which of the following statements is the reason for inaccurate information in the above situation? Group of answer...
-
Discounted Cash flow dan Non Discounted Cash flow. Jelaskan apa maksud dan perbedaan dari dua konsep tersebut. Metode kriteria analisis investasi apa saja yang termasuk dalam masing-masing konsep...
-
Bowling Green Financial is preparing its Budgeted Income Statement for next year. Bowling Green Financial has collected the following information about this statement: Sales for next year total...

Study smarter with the SolutionInn App


