Hydrazine (N 2 H 4 ) is used as a fuel in liquid-fueled rockets. When hydrazine reacts
Question:
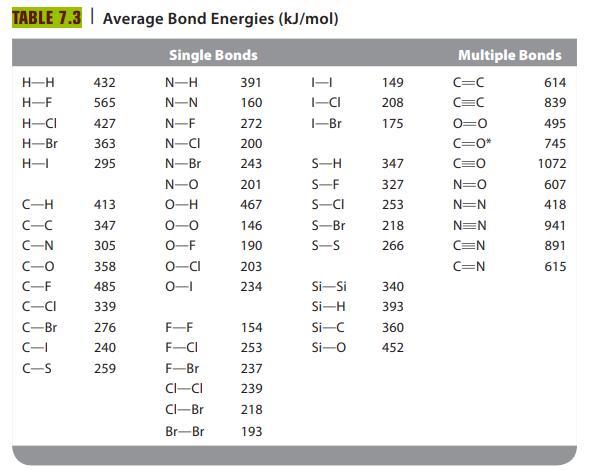
Hydrazine (N2H4) is used as a fuel in liquid-fueled rockets. When hydrazine reacts with oxygen gas, nitrogen gas and water vapor are produced. Write a balanced equation and use bond energies from Table 7.3 to estimate ΔH for this reaction.
Transcribed Image Text:
TABLE 7.3 Average Bond Energies (kJ/mol) Single Bonds N-H N-N H-H H-F H-CI H-Br H-1 C-H C-C C-N C-O C-F -CI C-Br C-I C-S 432 565 427 363 295 413 347 305 358 485 339 276 240 259 N-F N-CI N-Br N-O O-H 0-0 O-F O-CI 0-1 F-F F-CI F-Br CI-CI Cl-Br Br-Br 391 160 272 200 243 201 467 146 190 203 234 154 253 237 239 218 193 H I-CI 1-Br S-H S-F S-CI S-Bri S-S Si-Si Si-H Si-C Si-O 149 208 175 347 327 253 218 266 340 393 360 452 Multiple Bonds C=C C=C 0=0 C=O* C=0 N=O N=N N=N C=N C=N 614 839 495 745 1072 607 418 941 891 615
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The balanced chemical equation for the reaction between hydrazine N2H4 and oxygen ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write an equation for the reaction of hydrazine with fluorine gas to produce nitrogen gas and hydrogen fluoride gas. Estimate ÎH for this reaction, using bond energies from Table 13.6. Table...
-
A promising new material with great potential as a fuel in solid rocket motors is ammonium dinitramide [NH4N(NO2)2]. a. Draw Lewis structures (including resonance forms) for the dinitramide ion...
-
The steering rockets in space vehicles use N 2 O 4 and a derivative of hydrazine, 1,1-dimethylhydrazine (Study Question 5.86). This mixture is called a hypergolic fuel because it ignites when the...
-
Problem 3.6.1 Given the random variable Y in Problem 3.4.1, let U = g(Y) = Y2. (a) Find Pu(u). (b) Find Fu(u). (c) Find E[U].
-
Powderhorn Corporation reported sales of $257,000, net income of $45,300, cash of $9,300, and net cash provided by operating activities of $21,200. Accounts receivable have increased at three times...
-
Suppose that a producer in China is constrained by the MFA to sell a certain number of shirts, regardless of the type of shirt. For a T-shirt selling for $2.00 under free trade, the MFA quota leads...
-
The constant in Eq. (15-22b) is \(1.173 \times 10^{-16}\), which agrees with Geankoplis et al. (2018). However, Cussler (2009) and Wankat and Knaebel (2019) use a constant of 7.4 \(\times 10^{-8}\)....
-
Cost-plus target return on investment pricing. John Beck is the managing partner of a business that has just finished building a 60-room motel. Beck anticipates that he will rent these rooms for...
-
What is the average time a loaf of bread stays at Lulu's if the bakery sells 5 0 loaves per day and maintains an average inventory of 1 0 0 loaves?
-
Kimberly-Clark Corporation and Procter & Gamble Company reported the following information about inventory in their financial statements and footnotes. Use this information to answer the following...
-
Although nitrogen trifluoride (NF 3 ) is a thermally stable compound, nitrogen triiodide (NI 3 ) is known to be a highly explosive material. NI 3 can be synthesized according to the equation a. What...
-
Give the Lewis structure, molecular structure, and hybridization of the oxygen atom for OF 2 . Would you expect OF 2 to be a strong oxidizing agent like O 2 F 2 discussed in Exercise 61? Exercise 61....
-
Dr. Tooth is a 40-year-old dentist in Small Town, USA. He graduated from dental school five years ago and has had a thriving practice ever since. At the end of last year, however, Dr. Tooth had an...
-
Bheksizwe (Pty) Ltd is a company that specializes in the production of custom made mountain bikes, Bhekisizwe (Pty) Luis a micro bossiness and qualifies for all of the advantages of turnover tux. The...
-
1 If 83 cubic inches of O 2 gas at 56 psig is allowed to expand to 354 cubic inches w/o changing its temperature, what will be the final pressure of the O 2 gas expressed as a gauge pressure? 2. In a...
-
Tom spent his entire life in Australia until he ceased to be an Australian resident and became a resident of the United Kingdom in June 2 0 2 3 . When he left Australia, Tom s only asset was a...
-
11. As a hot hands pilot you get to fly an aircraft with air refueling capability. You take on fuel from a tanker and your weight before refueling was 30,000 lbs and you take on 5000 lbs of fuel. If...
-
Distinguish between personal selling and sales promotions and which you would use to increase sales of the following products. Products: Farm equipment Toyotas Calvin Klein
-
A confidence interval for a population mean is to be estimated. The population standard deviation is guessed to be anywhere from 14 to 24. The half-width B desired could be anywhere from 2 to 7. a....
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
Chemists recognize that the cyclohexyl radical is likely to be more stable than the cyclopentylmethyl radical, because they know that six-membered rings are more stable than five-membered rings and,...
-
The presence of the carbonyl group in a molecule is easily confirmed by an intense line in the infrared spectrum around 1700 cm 1 that corresponds to a CO stretching vibration. Locate this line in...
-
ge 25 40. Gloria's credit card charges an annual interest rate of 18.5% (0.05068% per day), compounded daily, on any outstanding balance. Her current outstanding balance is $2830.58. There is a...
-
Vance incorporated is considering investing in a project with the following expected cash flows: -186, 33, 44, 42. If Vance's expected cost of capital is 11.51%, what is the expected NPV of the...
-
Give me three reasons of why should invest in tesla according to the company activites ans goals and achievments in 2 0 2 3 - 2 0 2 4 Each reason should focus on only one specif goal

Study smarter with the SolutionInn App


