In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at
Question:
In the electrolysis of an aqueous solution of Na2SO4, what reactions occur at the anode and the cathode (assuming standard conditions)?
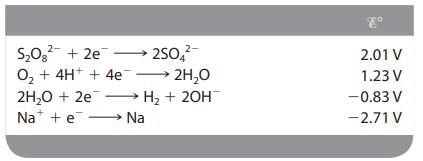
Transcribed Image Text:
2- S₂0² +2e 250 O₂ + 4H+ + 4e →→→ 2H₂O 2H₂O + 2e → H₂ + 2OH Na + e→→→→→→→→→ Na 2.01 V 1.23 V -0.83 V -2.71 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In the electrolysis of an aqu...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write equations for the half-reactions that occur at the anode and cathode in the electrolysis of molten KBr. What are the products formed at the anode and cathode in the electrolysis of aqueous KBr?
-
Which product, O 2 or F 2 , is more likely to form at the anode in the electrolysis of an aqueous solution of KF? Explain your reasoning.
-
Predict the products formed in the electrolysis of an aqueous solution of CdSO 4 .
-
Using Figure 7-5 as an example, redraw Figure 7-12 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
On October 5, Lane Company buys merchandise on account from OBrien Company. The selling price of the goods is $5,000, and the cost to OBrien Company is $3,000. On October 8, Lane returns defective...
-
Ranney Inc. has sales of $18,700, costs of $10,300, depreciation expense of $1,900, and interest expense of $1,250. If the tax rate is 40 percent, what is the operating cash flow?
-
The balance sheets of a business at 1 July 2008 and 30 June 2009 show net assets of 85,000 and 105,000 respectively. The profit for the year for this business is 15,000. The owner made regular cash...
-
Downs Company purchases a patent for $156,000 on January 2, 2014. Its estimated useful life is 6 years. (a) Prepare the journal entry to record amortization expense for the first year. (b) Show how...
-
In a random sample of 2,282 college students, 356 reported getting 8 or more hours of sleep per night. Create a 95% confidence interval for the proportion of college students who get 8 or more hours...
-
Lamson Sauces produces a hot sauce using tomatoes and chili peppers. Lamson developed the following standard cost sheet: On March 2, Lamson produced a batch of 32,000 ounces with the following actual...
-
Gold is produced electrochemically from an aqueous solution of Au(CN) 2 - containing an excess of CN - . Gold metal and oxygen gas are produced at the electrodes. What amount (moles) of O 2 will be...
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c. 0 d. HO e. C6H12O6 f. Ag g. PbSO4 h. PbO i. NaCO4 j. CO k. (NH4)2Ce(SO4)3 1. Cr03
-
Why is overhead typically estimated and allocated as opposed to just traced to each job and recorded as indirect costs are paid?
-
A ball with an initial velocity of 2.23 m/s rolls up a hill without slipping. (a) Treating the ball as a spherical shell, calculate the vertical height (in m) it reaches. 0.254 x m (b) Repeat the...
-
Consider a machine that costs $20,000 and has a five-year useful life. At the end of the five years, it can be sold for $4,000 after tax adjustment. The annual operating and maintenance (O&M) costs...
-
What is the role of cognitive reframing in fostering resilience, and how does it contribute to an individual's ability to navigate complex challenges ?
-
Fred Brookson, a lifelong resident of Klamath Falls, Oregon, has hired your firm to file suit against Wendell Carter for injuries sustained as a result of an incident that occurred in southern...
-
About a week before his performance review, Seth was asked to fill out a form that required him to list his strengths his weaknesses and his accomplishments work over the past few months Based on the...
-
A security service employing 10 officers has been asked to provide 3 persons for crowd control at a local carnival. In how many different ways can the firm staff this event?
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
Hydrocarbons are generally considered to be nonpolar or weakly polar at best, characterized by dipole moments that are typically only a few tenths of a debye. For comparison, dipole moments for...
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
QUESTION 9 Creative Sound Systems sold investments, land, and its own common stock for $30.0 million, $14.1 million, and $40.2 million, respectively. Creative Sound Systems also purchased treasury...
-
EcoMotorss disclosure notes for the year ending December 31, 2017, included the following regarding its $0.001 par common stock: Employee Stock Purchase Plan Our employees are eligible to purchase...
-
Exercise 7-10 (Algo) Customer Profitability Analysis [LO7-3, LO7-4, LO7-5] Worley Company buys surgical supplies from a variety of manufacturers and then resells and delivers these supplies to...

Study smarter with the SolutionInn App


