Order the following solids (ad) from least soluble to most soluble. Ignore any potential reactions of the
Question:
Order the following solids (a–d) from least soluble to most soluble. Ignore any potential reactions of the ions with water.
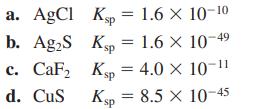
Transcribed Image Text:
a. AgCl Kp = 1.6 × 10-10 Ksp = 1.6 × 10- b. Ag₂S c. CaF₂ d. CuS Ksp = 4.0 × 10-¹¹ Ksp = 8.5 X 10-45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Note When a salt has a larger Ksp value its solubility will be hi...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Assuming that the solubility of Ca 3 (PO 4 ) 2 (s) is 1.6 10 -7 mol/L at 25 C, calculate the K sp for this salt. Ignore any potential reactions of the ions with water.
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
Perform the indicated operations and simplify the result. Leave your answer in factored form. X x - 3 x + 1 x2 + 5x 24
-
What type of bailment relationship was created when Denai agreed to store Finneys boat? What degree of care was Denai required to exercise in storing the boat? Vanessa Denai purchased forty acres of...
-
Using the information from Problem and the inventory information for Best Bikes below complete the requirements below. Assume income tax expense is $136,700 for the year. Inventories Raw materials,...
-
For Koren Company actual sales are \($1,200,000\) and break-even sales are \($900,000\). Compute (a) the margin of safety in dollars and (b) the margin of safety ratio.
-
Sweet Stores, Inc., sells electronics and appliances. The excerpts that follow are adapted from Sweet Stores' financial statements for 2016 and 2015? Requirements 1. How much was Sweet Stores' cost...
-
The following information was drawn from the balance sheets of the Kansas and Montana companies: Current assets Kansas $52,000 Montana Current liabilities $74,000 27,300 29,600 Required a. Compute...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
A 50.0-mL sample of 0.00200 M AgNO 3 is added to 50.0 mL of 0.0100 M NaIO 3 . What is the equilibrium concentration of Ag + in solution? (K sp for AgIO 3 is 3.0 10 -8 .)
-
Calculate the solubility of Co(OH) 2 (s) (K sp = 2.5 10 -16 ) in a buffered solution with a pH of 11.00.
-
Listed below are the time intervals (in minutes) before and after eruptions of the Old Faithful geyser. Find the values of d and s d . In general, what does ? d represent? Time interval before...
-
Discuss challenges to the implementation of a HIT plan for a healthcare organization which will effectively capture the important data points which can be used to monitor health outcomes. Provide...
-
Discuss the role of linear programming in project management and how it can be used to optimize resource allocation and scheduling.
-
The function f(x) = 2x - x2 is given graphed below: Note: Click on graph for larger version in new browser window. (A) Starting with the formula for f(x), find a formula for g(x), which is graphed...
-
OA solid object has an apparent weight 15.2 N when completely submerged in ethyl alcohol (density 790) and 13.7 N when completely submerged in water. Find the volume of the object.
-
What is Business Management? Business Management is the process of developing and executing a plan to achieve objectives and success in an organization.
-
The table shows results of whether the death penalty was imposed in murder trials in Florida between 1976 and 1987. For instance, the death penalty was given in 53 out of 467 cases in which a white...
-
Wimot Trucking Corporation uses the units-of-production depreciation method because units-of-production best measures wear and tear on the trucks. Consider these facts about one Mack truck in the...
-
Identify the reagents that you would use to accomplish each of the following transformations: (a) (b) H.
-
Predict the major product for each of the following transformations: (a)
-
Identify the reagents that you would use to accomplish each of the following transformations (you will also need to use reactions from previous chapters). (a) (b) (c) Br Br OH
-
What is the main purpose of a photographer?Select one:a.To write a script for the show.b.To finance and handle the production and cost needed in order to publish a print material.c.To capture images...
-
Accounting I15) SO ICEY Inc. has a periodic inventory system. The company purchased 225 units ofinventory at $11.50 per unit and 350 units at $12.50 per unit. What is the weightedaverage unit cost...
-
6-9Unearned Income of Minor Children and Certain Students (LO 6.4)Explain the two different ways that the tax on unearned income of minor children, or "kiddie tax," can be reported.The "kiddie tax"...

Study smarter with the SolutionInn App


