The Hg 2+ ion forms complex ions with I - as follows: A solution is prepared by
Question:
The Hg2+ ion forms complex ions with I- as follows:
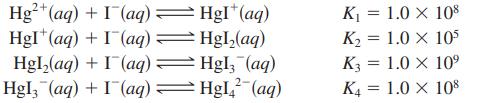
A solution is prepared by dissolving 0.088 mole of Hg(NO3)2 and 5.00 moles of NaI in enough water to make 1.0 L of solution.
![a. Calculate the equilibrium concentration of [HgI4]. b. Calculate the equilibrium concentration of [I]. c.](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/9/6/4/82265536796b3c181699964823549.jpg)
Transcribed Image Text:
Hg²+ (aq) + I (aq) = HgI+ (aq) + (aq) = (aq): Hgl₂(aq) + Hgl, (aq) + (aq) → = HgI*(aq) Hgl₂(aq) Hgl, (aq) HgI₂² (aq) K₁ = 1.0 X 108 K₂ = 1.0 X 105 K3 = 1.0 X 109 K4 1.0 X 108
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The Hg2 ion forms complex ions with I as follows Hg2 I HgI HgI I HgI2 HgI2 I HgI3 HgI3 I HgI4 The eq...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
A solution is prepared by dissolving table salt, sodium chloride, in water at room temperature. a. Assuming there is no significant change in the volume of water during the preparation of the...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A solution is prepared by dissolving 10.8 g ammonium sulfate in enough water to make 100.0 mL of stock solution. A 10.00-mL sample of this stock solution is added to 50.00 mL of water. Calculate the...
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
Curtis is an executive on a business trip to the West Coast. He has driven his car on this trip and checks into the Hotel Ritz. The hotel has a guarded underground parking lot. Curtis gives his car...
-
he times required to complete each of five jobs in a two-machine flow strop are shown in the following table. Each job must follow the same sequence, begin with machine A and end with machine B. a....
-
Sales of automotive products for Ford Motor Company and General Motors Corporation for a five-year period are: Net sales for Pfizer Inc. and Abbott Laboratories for the same five years follow:...
-
Data on U.S. work-related fatalities by cause follow (The World Almanac, 2012). Cause of Fatality Number of Fatalities Transportation incidents ............. 1795 Assaults and violent acts...
-
Lynette, a supervisor, gives average ratings to all of her direct reports regardless of their actual level of performance. This is an example of which rating error? Lynette, a supervisor, gives...
-
Suppose that Sasha and Jayla both collect data to investigate whether people tend to call heads more often than tails when they are asked to call the result of a coin flip. If Sasha has a smaller...
-
Calculate the mass of manganese hydroxide present in 1300 mL of a saturated manganese hydroxide solution. For Mn(OH)2, K sp = 2.0 10 -13 .
-
Write equations for the stepwise formation of each of the following complex ions. a. Ni(CN) 4 2- b. V(C2O 4 ) 3 3 -
-
For the following exercises, find the inverse of the functions. f(x) = 4 2x 3
-
How do you change your default shell to C shell (csh) ?
-
Needs Analysis : The analysis of the information gathered on the community military veterans and mental health. This will provide a conclusions you have drawn from thinking about the findings you...
-
How should an organization deal with third-party network access?
-
PAYROLL Using the data provided, complete the payroll calculations table. Download the deductions table from Accounting Supplemental Material Page. Submit your reconciliation in Accounting Competency...
-
Explain your personal learning goals for this course. Be sure to identify at least two areas in which you feel you would like to deepen your current level of understanding of Leadership and Emerging...
-
The following transactions occurred during the month for Tori Peel, CPA: Jun. 1 Peel opened an accounting firm by contributing $13,500 cash and office furniture with a fair market value of $5,400 in...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
Distinguish between the following terms applied to a set of functions: orthogonal and normalized, and orthonormal.
-
Why can we conclude that the wave function (x, t) = (x) e i (E / h )t represents a standing wave?
-
What is the usefulness of a complete set of functions?
-
a. Given the following holding-period returns, E. compute the average returns and the standard deviations for the Zemin Corporation and for the market. b. If Zemin's beta is 1.28 and the risk-free...
-
Students will analyze the accounting and finance employment and careers environment, identify a specific role and relate it to their personal career plan and prepare: Part A. A written response that...
-
Weekly demand for boxes follows a normal distribution with mean 448 and standard deviation 64. Each week, a replenishment order is placed with its supplier and the order arrives 4 weeks later. All...

Study smarter with the SolutionInn App


