Explain why benzenesulfonic acid is a Brnsted acid. H-0: :0-S :0: benzenesulfonic acid
Question:
Explain why benzenesulfonic acid is a Brønsted acid.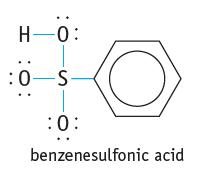
Transcribed Image Text:
H-0: :0-S :0: benzenesulfonic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Benzenesulfonic acid is a Brnsted acid because it can donate a proton H to another substa...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Work Problem 3.27 for the reverse reactions in Fig. P3.27 Problem 3.27 (a) Classify each reaction step in the forward direction with one or more of the following terms: (1) A Lewis acid-base reaction...
-
Benzenesulfonic acid is a monoprotic acid with pKa = 2.25. Calculate the pH of a buffer composed of 0.150 Mbenzenesulfonic acid and 0.125 M sodium benzenesulfonate.
-
Write a stepwise mechanism for the hydrolysis of chlorobenzene under the conditions shown in Table 24.3. TABLE 24.3 Industrial Syntheses of Phenol Reaction and comments Chemical equation Reaction of...
-
Since the early 2000s, there has been a significant increase in the price of corn-based ethanol. a. A key input in the production of corn-based ethanol is corn. Use an appropriate diagram to explain...
-
In the past decade, the Japanese economy has fallen from its lofty levels of the 1980s. As a consequence, many Japanese companies have been forced to downsize. In most companies, one of two...
-
Yang Co. was organized on April 1, 2017. The company prepares quarterly financial statements. The adjusted trial balance amounts at June 30 are shown below. (a) Determine the net income for the...
-
What term is used to describe products that are being manufactured but are not yet complete?
-
The balance sheet for Stud Clothiers is shown next. Sales for the year were $2,400,000, with 90 percent of sales sold on credit. Compute the following ratios: a. Current ratio. b. Quick ratio. c....
-
Assume you own and run a small ice cream shop located on the grounds of a private pool. You want to maximize sales and decide that allowing customers to buy on credit could be a big driver of sales...
-
m-Nitrophenol, a weak acid, can be used as a pH indicator because it is yellow at a pH above 8.6 and colorless at a pH below 6.8. If the pH of a 0.010 M solution of the compound is 3.44, calculate...
-
The chapter opening photograph (page 670) showed how the cobalt(II) chloride equilibrium responded to temperature changes. (a) Look back at that photograph. Is the conversion of the red cation to the...
-
For the van der Waals interaction with potential-energy function given by Eq. (13.25), show that when the magnitude of the displacement x from equilibrium (r = Ro) is small, the potential energy can...
-
A family amortizes a mortgage of $420, 000 with monthly payments of j2 = 3.2% for 22 years. Determine the final payment made. A reminder that marks are only given for work shown. If done by EXCEL, no...
-
Assume we have a stock currently worth $50. We also assume the interest rate is zero, and we can buy options for this stock with a strike price of $50. If the stock can rise or fall by $10 with equal...
-
Explain any five distinction between a Mutual Fund Company and a Venture Capital Company 2. How do you differentiate the primary market from the secondary market? ( List out any five distinction...
-
A firm has a return on equity of 10%, Earnings per share of $2 and is expected to pay $0.5 per share annual dividend. If you require 12.5% rate of return, how much are you willing to pay for this...
-
A. Identify the different types of yield curves and explain what they indicate for the U.S economy? What is the current shape of the yield curve and why is it shaped that way?(Something short just to...
-
A tax planner for a start up biotechnology company is advising her client about how to efficiently organize R& D activities. One suggestion the tax planner made is to form a joint venture with...
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
A certain metal surface emits electrons when light is shone on it. (a) How can the number of electrons per second be increased? (b) How can the energies of the electrons be increased?
-
Would you expect the gravitational attractive force between two protons in a nucleus to counterbalance their electrical repulsion? Calculate the ratio between the electric and gravitational forces...
-
Which constituents of an atom consist of quarks and which do not?
-
ped Saving for College Monthly (End and Beg) The parents of Mr. and Mrs. Smith have decided to give their newborn grandchild $177 at the end of each month for the next 18 years to help pay for...
-
Your uncle has agreed to deposit $8035 in your brokerage account at the beginning of each of the four years (t=0, t=1, t=2, t=3).You estimate that you can earn 7 percent a year on your...
-
What type of probability distribution will Elva need to use to generate random observations for delivery times so that she can set up the drivers' schedules

Study smarter with the SolutionInn App


