In the presence of oxgyen and acid, two half-reactions responsible for the corrosion of iron are Calculate
Question:
In the presence of oxgyen and acid, two half-reactions responsible for the corrosion of iron are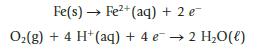
Calculate the the standard potential, E°, and decide whether the reaction is product-favored at equilibrium. Will decreasing the pH make the reaction less thermodynamically product-favored at equilibrium?
Transcribed Image Text:
Fe(s)→ Fe²+ (aq) + 2 e O₂(g) + 4 H+ (aq) + 4 e → 2 H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Calculation of the standard potential E The standard potential of a reaction is calculated by subtra...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
In the presence of the enzyme aconitase, the double bond of aconitic acid undergoes hydration. The reaction is reversible, and the following equilibrium is established: (a) The major tricarboxylic...
-
In a study to compare two different corrosion inhibitors, specimens of stainless steel were immersed for four hours in a solution containing sulfuric acid and a corrosion inhibitor. Forty-seven...
-
In the presence of oxygen and water, two half-reactions responsible for the corrosion of iron are Calculate the the standard potential, E, and decide whether the reaction is product-favored at...
-
If the appropriate discount rate for the following cash flows is 7.13 percent per year, what is the present value of the cash flows? Year Cash Flow 1 ......................$1,400 2...
-
Dayton Industrial produces a variety of chemicals that are used in an array of commercial applications. One popular product, a chemical solvent, contains two very caustic acids, A and B, each of...
-
The trial balance columns of the worksheet for Warren Roofing at March 31, 2017, are as follows. Other data: 1. A physical count reveals only $480 of roofing supplies on hand. 2. Depreciation for...
-
Johnson & Johnson discloses in its annual report that costs associated with advertising are expensed in the year incurred and are included in selling, marketing and administrative expenses on the...
-
Ken Jones, an architect, organized Jones Architects on April 1, 2014. During the month, Jones Architects completed the following transactions: a. Transferred cash from a personal bank account to an...
-
6) If the coupon rate equals the required rate of return, the price of the bond a. should be above its par value. b. should be below its par value. c. should be equal to its par value. d. is...
-
Balance the following equations. (a) Zn(s) + VO+ (aq) Zn+ (aq) + V+ (aq) (b) Zn(s) + VO3(aq) V2+ (aq) + Zn+ (aq) (c) Zn(s) + CIO- (aq) Zn(OH)(s) + Cl(aq) (d) CIO- (aq) + [Cr(OH)4](aq) Cl(aq) +...
-
Use E values to predict which of the following metals, if coated on nickel, will provide cathodic protection against corrosion to nickel. (a) Cu (b) Mg (c) Zn (d) Cr
-
A cylinder fitted with a piston has an initial volume of 0.1 m3 and contains nitrogen at 100 kPa, 25oC. The piston is moved, compressing the nitrogen until the pressure is 1.5 MPa and the temperature...
-
A local barbershop cuts the hair of 1,300 customers per month. The clients are men, and the barbers offer no special styling. During the month of May, 1,300 customers were serviced. The cost of...
-
Explain the positive and negative aspects of speculation in financial markets.
-
what is a reason for using just one rate , a long term yield as the average cost of debt financing for a firm that has multiple issues of debt wtih varyiny maturities?
-
What recommendations can help her organize ideas in planning stage old writing step?
-
Based on analysis, deck-building business is best Only consider what is an added cost and different from current business, if shifting things from current business to new then do not include
-
How difficult is it in reality to compute the corporations marginal tax rate? Why? What are the factors that are really important? If we observe that a firm has net operating losses, does this mean...
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
Draw an SM chart for the square root circuit of Problem 4.14. Data from Problem 4.14. This problem involves the design of a circuit that finds the square root of an 8-bit unsigned binary number N...
-
Draw an SM chart for the binary multiplier of Problem 4.22. Data from Problem 4.22. Design a multiplier that will multiply two 16-bit signed binary integers to give a 32-bit product. Negative numbers...
-
Design a binary-to-BCD converter that converts a 10-bit binary number to a 3-digit BCD number. Assume that the binary number is 999. Initially the binary number is placed in register B. When an St...
-
Supply chain overview: InnoMedRepairCo (deliver) -> (source) Retailer (like SuccessfulLivingCo) (deliver) -> Consumers InnoMedRepairCo sells the repaired devices to large numbers of online small...
-
Analyzing the presented data on an overall basis first, it is clear that Metropolitan Nashville General Hospital (MNGH) and Palestichi Clinic for People (PCP) have both parallels and variances in...
-
A modern industry that may end up falling afoul of antitrust law is ? A . insurance B . Healthcare C . Banking D . Technology

Study smarter with the SolutionInn App


