The boron trihalides (except BF 3 ) hydrolyze completely to boric acid and the acid HX. (a)
Question:
The boron trihalides (except BF3) hydrolyze completely to boric acid and the acid HX.
(a) Write a balanced equation for the reaction of BCl3 with water.
(b) Calculate ΔrH° for the hydrolysis of BCl3 using data in Appendix L and the following information:
ΔfH° [BCl3(g)] = −403 kJ/mol; ΔfH° [B(OH)3(s)]= −1094 kJ/mol.
Data given in Appendix L
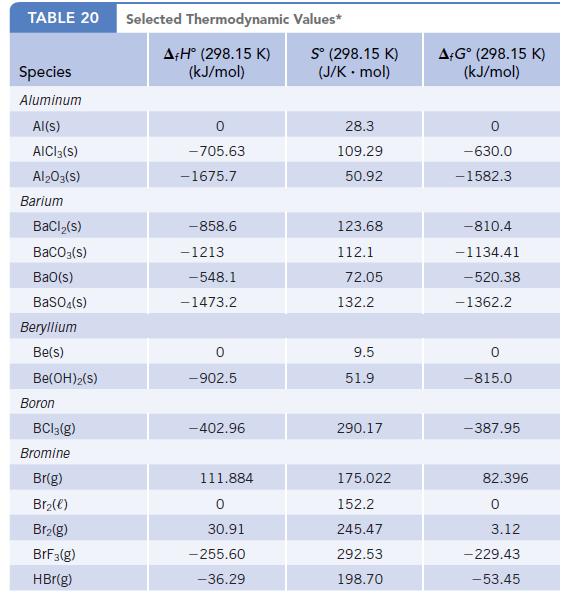
Transcribed Image Text:
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -705.63 -1675.7 -858.6 -1213 -548.1 -1473.2 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K . mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 A+Gᵒ (298.15 K) (kJ/mol) -630.0 -1582.3 -810.4 -1134.41 -520.38 -1362.2 0 -815.0 -387.95 82.396 0 3.12 - 229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Balanced chemical reaction for BCI and water B...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Boron nitride (BN) exists in two forms. The first is a slippery solid formed from the reaction of BCl3 with NH3, followed by heating in an ammonia atmosphere at 750oC. Subjecting the first form of BN...
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
Write a balanced equation for the complete oxidation of each of the following, and calculate the respiratory quotient for each substance. (a) Ethanol (b) Acetic acid (c) Stearic acid (d) Oleic acid...
-
Craig's Bowling Inc. operates several bowling centres (for games and equipment sales). The following transactions occurred in November 2017. For each of the following transactions, complete the...
-
Assume that two kinds of buyers purchase contracts from a monopolist who promises to deliver goods in the future. One kind of buyer values the good more highly than the other. The monopolist would...
-
Can you explain why many of the forces you named in the answer to the previous question have led to significant problems for the management of banks and other financial firms and for their...
-
What are the factors that explain the difference in yields to maturity between long-term and short-term bonds?
-
The profit function for two products is Profit = -3x12 + 42x1 3x22 + 48x2 + 700 Where x1 represents units of production of product 1 and x2 represents units of production of product 2. Producing one...
-
You are analyzing the cost of debt for a firm. You know that the firm's 14-year maturity, 10.75 percent coupon bonds are selling at a price of $901.34. The bonds pay interest semiannually. If these...
-
Draw a possible structure for the cyclic anion in the salt K 3 B 3 O 6 and the anion in Ca 2 B 2 O 5 .
-
The element below aluminum in Group 3A is gallium, and there are numerous similarities in the chemistry of these two elements. For example, the hydroxides of both elements are amphoteric. A...
-
1. If money is not backed by gold, what gives it its value? 2. If the money that banks have created vastly exceeds the amount of cash in the economy, does this mean that there is too much money in...
-
Explaining the bullet points for the "The Great Travel Hack" below is the link to the video. https://vimeo.com/328574477 Find an episode of "The Great Travel Hack" campaign by Shell in the reading...
-
In what ways does the phenomenon of "emotional contagion" permeate organizational dynamics, influencing the socio-emotional climate, and subsequently impacting employee morale, job satisfaction, and...
-
In your personal finance journal, record your bond strategy. What will be your purpose in including bonds in your portfolio? What types of bonds will you include and why? Will you take an active or...
-
Titanic Corporation's net income for the year ended December 31, 2022, is $380,000. On June 30, 2022, a $0.75 per-share cash dividend was declared for all common shareholders. Outstanding at the time...
-
An open box with a rectangular base is to be made from a piece of tin measuring 30 cm by 45 cm by cutting out a square from each corner and turning up the sides. Find the dimensions that yield the...
-
Nine homes are chosen at random from real estate listings in two suburban neighborhoods, and the square footage of each home is noted in the following table. At the .10 level of significance, is...
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
Figure 2.24 (page 39) shows one of Galileos experiments in which a ball rolls up an incline. A ball that is initially rolling up the incline will roll up to some maximum height and then roll back...
-
A rabbit runs in a straight line with a velocity of +1.5 m/s for a period of time, rests for 10 s, and then runs again along the same line at +0.60 m/s for an unknown amount of time. The rabbit...
-
Figure P2.36 shows the acceleration as a function of time for an object. (a) If the object starts from rest at t - 0, what is the velocity of the object as a function of time? (b) If the object...
-
Bird's Eye Treehouses, Incorporated, a Kentucky company, has determined that a majority of its customers are located in the Pennsylvania area. Therefore, it is considering using a lockbox system...
-
Given a binomial random variable with n =100 and p =0.4,estimate the Pr[X30] Please send me answer in type form strictly prohibited handwritten solution and don't use chatgpt otherwise I will report...
-
Specific audit objectives in this area include ascertaining that Payroll charges, materials, and other direct costs are summarized accurately and distributed to individual contracts or grant...

Study smarter with the SolutionInn App


