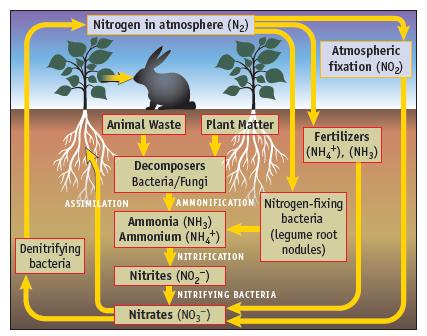
The nitrogen cycle (Figure 20.1) shows the oxidation of NH 4 + , first to NO 2
Question:
The nitrogen cycle (Figure 20.1) shows the oxidation of NH4+, first to NO2− and then the subsequent oxidation of NO2− to NO3−. Write balanced equations for each of these half-reactions (in acid solution).
Data given in Figure 20.1
Transcribed Image Text:
Nitrogen in atmosphere (N₂) Denitrifying bacteria Animal Waste Plant Matter ASSIMILATION Decomposers Bacteria/Fungi Ammonia (NH3) Ammonium (NH4+) AMMONIFICATION Nitrogen-fixing bacteria NITRIFICATION Nitrites (NO₂) Atmospheric fixation (NO₂) NITRIFYING BACTERIA Nitrates (NO3) Fertilizers (NH,*), (NH3) (legume root nodules)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The following are balanced equations for the oxidation of NH4 to NO2 and the oxidation of NO2 to NO3 ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
Write balanced equations for the reactions described in Table 18.13 for the production of Bi and Sb. Table 18.13 Element Nitrogen Phosphorus Electronegativity Source Method of Preparation 3.0 2.2 Air...
-
Write balanced equations for each of the following. a. Solid calcium fluoride is heated with sulfuric acid to give hydrogen fluoride vapor. b. Solid potassium chlorate is carefully heated to yield...
-
Can you give us a few Google AdWords Ad Extensions names that you know?
-
Integrated Technologies Inc. is considering the purchase of automated machinery that is expected to have a useful life of four years and no residual value. The average rate of return on the average...
-
Which of the following transactions would be considered noncash investing and financing activities? _______ 1. Additional borrowing from bank. _______ 2. Purchase of equipment with investments....
-
Professional practice models allow for autonomous nursing practice. This concept is based on the relationship between the worker and the workplace. An example of a professional practice model is: A....
-
Selected financial data of two competitors, Blockbuster Inc. and Movie Gallery, Inc., in a recent year are presented below and on page 91. (All dollars are in millions.) InstructionsFor each company,...
-
Can I know how to answer question 6 )a ,b and c
-
Refer to the figure below and Figure 20.25, which show the fraction of species in solution [alpha ()] as a function of pH. The following questions are in regard to the equilibria involved in an...
-
The refrigerating liquids in air conditioners and refrigerators are largely chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs). Among the latter family of compounds is the refrigerant...
-
Who prescribes GAAP for U.S. companies?
-
Bryant Company reports net income of $22,200. For the year, depreciation expense is $9,200 and the company reports a gain of $5,200 from sale of machinery. It also had a $4,200 loss from retirement...
-
Regarding the diagnostic/ analytic models appropriate for analyzing the data, either an individual-level diagnostic model or an organization-level diagnostic model would work. Briefly explain how...
-
During October, Rover Industries produced 35,300 units of product with costs as follows: Direct materials$85,500 Direct labor43,750 Variable overhead14,500 Fixed overhead150,000 Total: $293,750 What...
-
(16 points) The stock of Line of Duty (LoD), of which you own 645 shares, will pay a 3-per-share dividend one year from today. Two years from now, LoD will close its doors; stockholders will receive...
-
When converting a traditional sequential program to make it run as a parallelizable program, you need to consider four things as Partitioning, Communication, Synchronization, and Load balancing....
-
Suppose you are an employee of HP Corporation. You face a personal marginal tax rate on ordinary income of 50%, and on capital gains the tax rate is 20%. Your after-tax opportunity cost of capital is...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Derive the state transition table and D flip-flop input equations for a counter that counts from 1 to 6 (and back to 1 and continues).
-
Reduce the following state table to a minimum number of states. Present State Next State Output X = 1 X = 0 X= 1 B A D D G D
-
A Mealy sequential circuit is implemented using the circuit shown in Problem 1.26. Assume that if the input X changes, it changes at the same time as the falling edge of the clock. (a) Complete the...
-
content area top Part 1 Is the study experimental or observational? A stock analyst selects a stock from a group of twenty for investment by choosing the stock with the greatest earnings per share...
-
A card is being taken out one by one without replacement from a deck of well-shuffled cards till we obtain an ace of spades. The probability of obtaining an ace of spades by the tenth draw is given...
-
Please answers these question with excel : Note. Usea = 0.05 for all tests. The Bank Call Center data shows the data on 70 former employees at one of thebank's call centers. a. Is the average length...

Study smarter with the SolutionInn App


